Gout is a common type of arthritis that causes intense pain, swelling, and stiffness in a joint. It usually affects the joint in the big toe.
Gout attacks can come on quickly and keep returning over time, slowly harming tissues in the region of the inflammation, and can be extremely painful. Hypertension, cardiovascular, and obesityare risk factors for gout.
It is the most common form of inflammatory arthritis in men, and although it is more likely to affect men, women become more susceptible to it after the menopause.
The Centers for Disease Control and Prevention (CDC) report that 8.3 million Americans were affected by gout between 2007 to 2008.
Fast facts on goutHere are some key points about gout. More detail and supporting information is in the main article.
- Gout is a form of arthritis caused by excess uric acid in the bloodstream.
- The symptoms of gout are due to the formation of uric acid crystals in the joints and the body’s response to them.
- Gout most classically affects the joint in the base of the big toe.
- Gout attacks often occur without warning in the middle of the night.
- Most gout cases are treated with specific medications.
Treatment
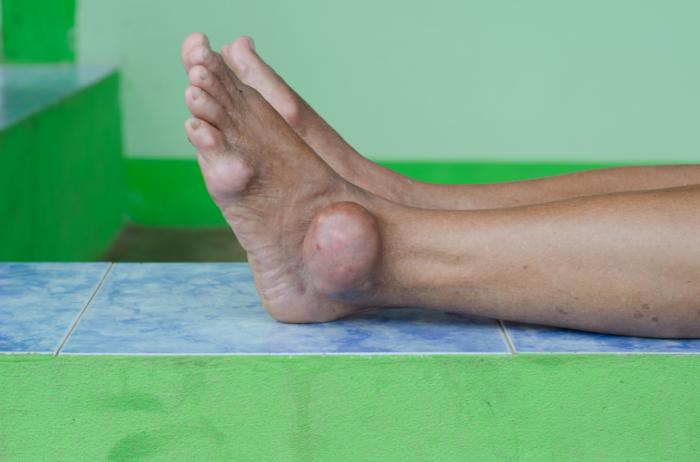
Gout patients often have acute inflammation around their joints.
The majority of gout cases are treated with medication. Medication can be used to treat the symptoms of gout attacks, prevent future flares, and reduce the risk of gout complications such as kidney stones and the development of tophi.
Commonly used medications include nonsteroidal anti-inflammatory drugs (NSAIDs), colchicine, or corticosteroids. These reduce inflammation and pain in the areas affected by gout and are usually taken orally.
Medications can also be used to either reduce the production of uric acid (xanthine oxidase inhibitors such as allopurinol) or improve the kidney’s ability to remove uric acid from the body (probenecid).
Without treatment, an acute gout attack will be at its worst between 12 and 24 hours after it began. A person can expect to recover within 1 to 2 weeks without treatment, but there may be significant pain during this period.
Tests and diagnosis
Gout can be tricky to diagnose, as its symptoms, when they do appear, are similar to those of other conditions. While hyperuricemia occurs in the majority of people that develop gout, it may not be present during a flare. On top of that, the majority of people with hyperuricemia do not develop gout.
One diagnostic test that doctors can carry out is the joint fluid test, where fluid is extracted from the affected joint with a needle. The fluid is then examined to see if any urate crystals are present.
As joint infections can also cause similar symptoms to gout, a doctor can look for bacteria when carrying out a joint fluid test in order to rule a bacterial cause. The fluid may need to be sent to a lab, where it can take several days to analyze.
Doctors can also do a blood test to measure the levels of uric acid in the blood, but, as mentioned, people with high uric acid levels do not always experience gout. Equally, some people can develop the symptoms of gout without having increased levels of uric acid in the blood.
Finally, doctors can search for urate crystals around joints or within a tophus using ultrasound scan. X-rays cannot detect gout, but may be used to rule out other causes.
Types
There are various stages through which gout progresses, and these are sometimes referred to as different types of gout.
Asymptomatic hyperuricemia
It is possible for a person to have elevated uric acid levels without any outward symptoms. At this stage, treatment is not required, though urate crystals may deposit in tissue and cause slight damage.
People with asymptomatic hyperuricemia may be advised to take steps to address any possible factors contributing to uric acid build-up.
Acute gout
This stage occurs when the urate crystals that have been deposited suddenly cause acute inflammation and intense pain. This sudden attack is referred to as a “flare” and will normally subside within 3 to 10 days. Flares can sometimes be triggered by stressful events, alcohol and drugs, as well as cold weather.
Interval or intercritical gout
This stage is the period in between attacks of acute gout. Subsequent flares may not occur for months or years, though if not treated, over time, they can last longer and occur more frequently. During this interval, further urate crystals are being deposited in tissue.
Chronic tophaceous gout
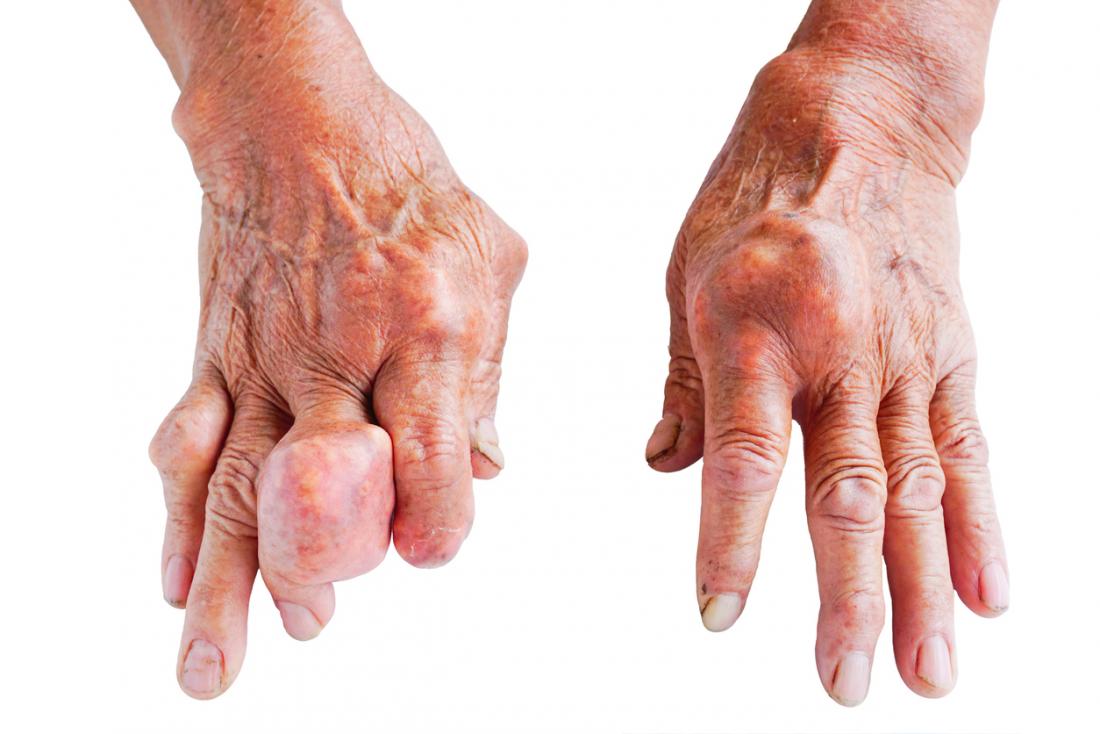
Chronic tophaceous gout is the most debilitating type of gout. Permanent damage may have occurred in the joints and the kidneys. The patient can suffer from chronic arthritis and develop tophi, big lumps of urate crystals, in cooler areas of the body such as the joints of the fingers.
It takes a long time without treatment to reach the stage of chronic tophaceous gout – around 10 years. It is very unlikely that a patient receiving proper treatment would progress to this stage.
Pseudogout
One condition that is easily confused with gout is pseudogout. The symptoms of pseudogout are very similar to those of gout, although thr flare-ups are usually less severe.
The major difference between gout and pseudogout is that the joints are irritated by calciumpyrophosphate crystals rather than urate crystals. Pseudogout requires different treatment to gout.
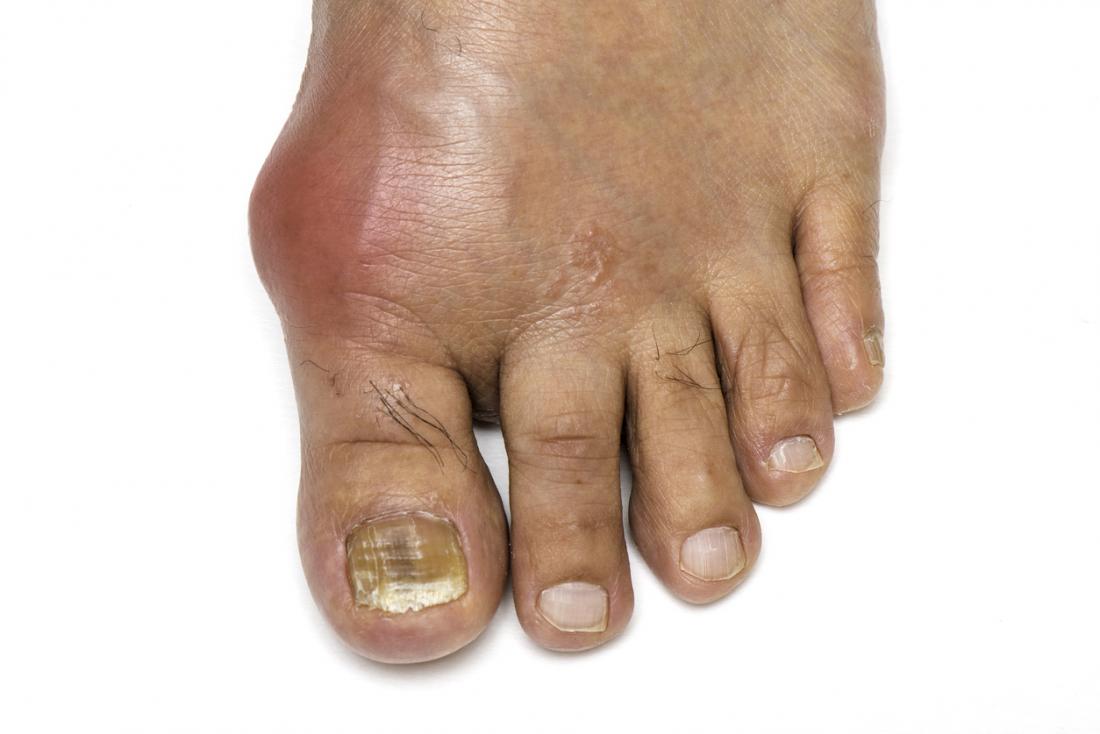
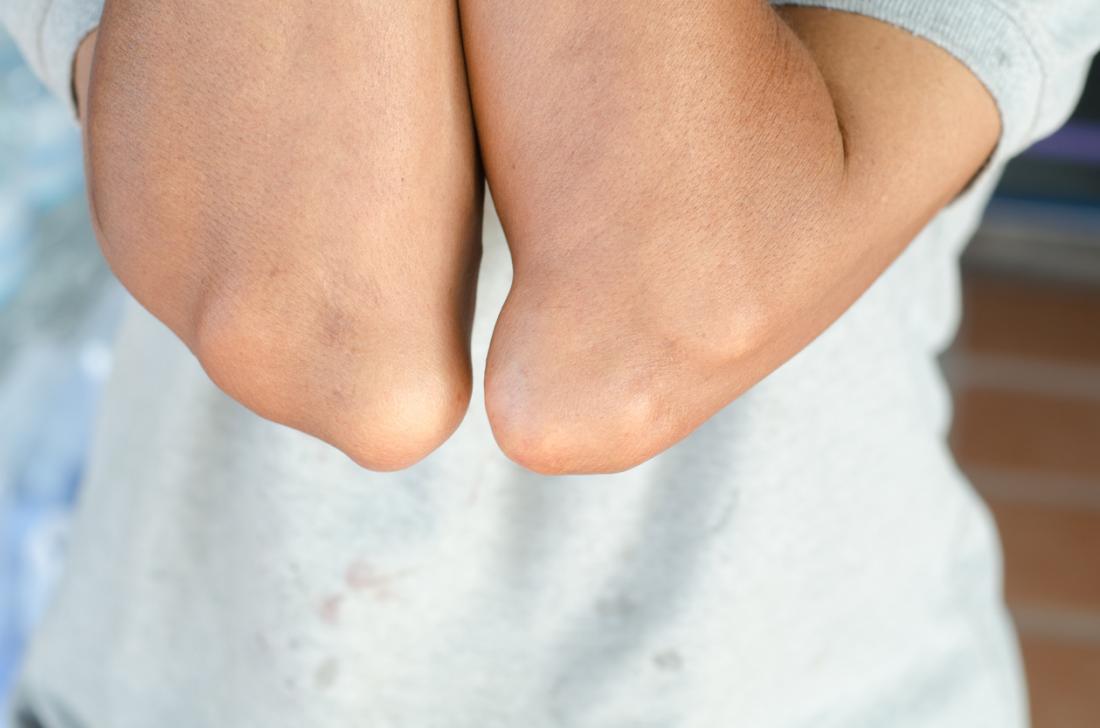
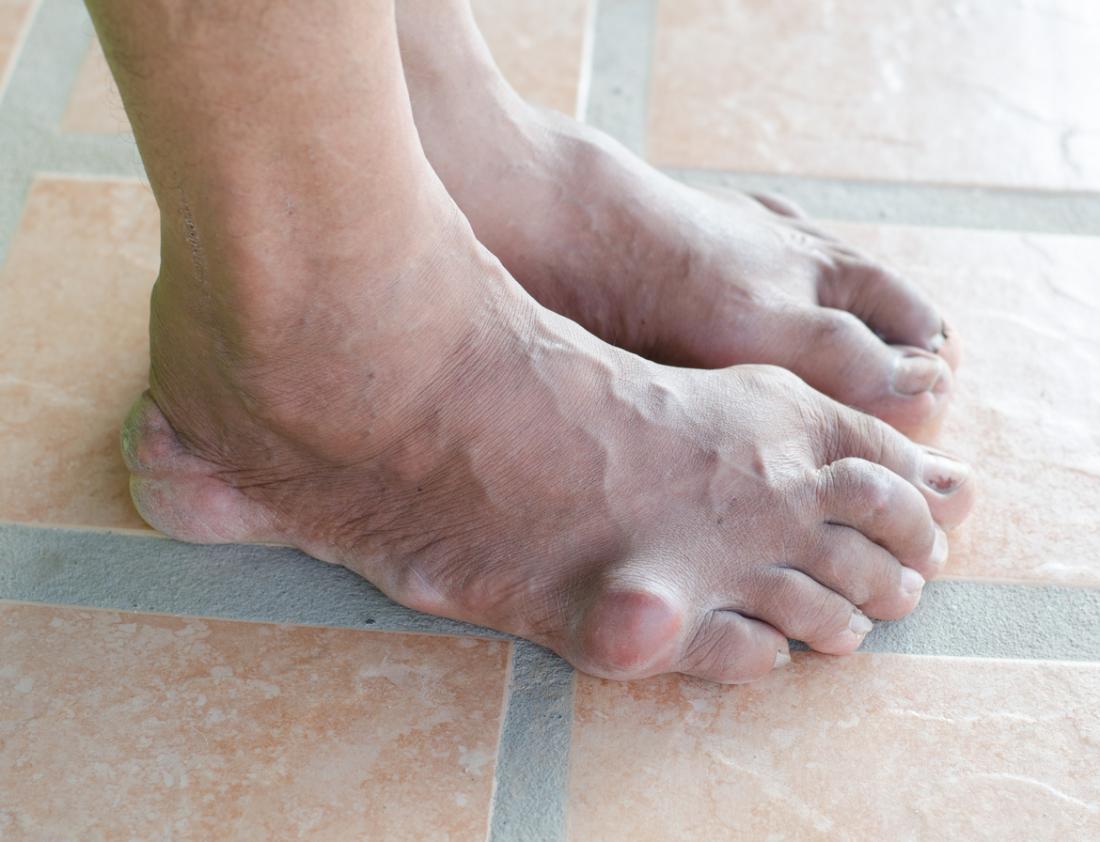
Pictures
The following images show the appearance of gout when it develops.
Causes
Gout is caused initially by an excess of uric acid in the blood, or hyperuricemia. Uric acid is produced in the body during the breakdown of purines – chemical compounds that are found in high amounts in certain foods such as meat, poultry, and seafood.
Normally, uric acid is dissolved in the blood and is excreted from the body in urine via the kidneys. If too much uric acid is produced, or not enough is excreted, it can build up and form needle-like crystals that trigger inflammation and pain in the joints and surrounding tissue.
Risk factors
There are a number of factors that can increase the likelihood of hyperuricemia, and therefore gout:
Age and gender: Men produce more uric acid than women, though women’s levels of uric acid approach those of men after the menopause.
Genetics: A family history of gout increases the likelihood of the condition developing.
Lifestyle choices: Alcohol consumption interferes with the removal of uric acid from the body. Eating a high-purine diet also increases the amount of uric acid in the body.
Lead exposure: Chronic lead exposure has been linked to some cases of gout.
Medications: Certain medications can increase the levels of uric acid in the body; these include some diuretics and drugs containing salicylate.
Weight: Being overweight increases the risk of gout as there is more turnover of body tissue, which means more production of uric acid as a metabolic waste product. Higher levels of body fat also increase levels of systemic inflammation as fat cells produce pro-inflammatory cytokines.
Recent trauma or surgery: Increases risk.
Other health problems: Renal insufficiency and other kidney problems can reduce the body’s ability to efficiently remove waste products, leading to elevated uric acid levels. Other conditions associated with gout include high blood pressure and diabetes.
Symptoms
Gout usually becomes symptomatic suddenly without warning, often in the middle of the night.
The main symptoms are intense joint pain that subsides to discomfort, inflammation, and redness.
Gout frequently affects the large joint of the big toe, but can also affect the forefoot, ankles, knees, elbows, wrists, and fingers.
The pain can be excruciating. A veteran visiting a Hospital in Birmingham, AL, said:
“I’ve been shot, beat up, stabbed, and thrown out of a helicopter, but none of that compared to the gout.”
Complications
In some cases, gout can develop into more serious conditions, such as:
- Kidney stones: If urate crystals collect in the urinary tract, they can become kidney stones.
- Recurrent gout: Some people only ever have one flare up; others may have regular recurrences, causing gradual damage to the joints and surrounding tissue.
Prevention tips
There are many lifestyle and dietary guidelines that can be tried to protect against flares or prevent gout from occurring in the first instance:
- maintain a high fluid intake of around 2 to 4 liters a day
- avoid alcohol
- maintain a healthy body weight
Home remedies
Individuals with gout can manage flare-ups by moderating their diet. A balanced diet can help reduce symptoms.
Decreasing foods that are high in purines, to ensure that the levels of uric acid in the blood do not get too high, is reasonable to try. Here is a list of high-purine foods to be wary of:
- anchovies
- asparagus
- beef kidneys
- brains
- dried beans and peas
- game meats
- gravy
- herring
- liver
- mackerel
- mushrooms
- sardines
- scallops
- sweetbreads
While it is reasonable to decrease or avoid these foods, it has been found that a high purine-rich diet does not increase the risk of gout, or aggrevate symptoms in research studies.
Asparagus, beans, some other plant-based foods, and mushrooms are also sources of purines, but research suggests that these do not trigger gout attacks and do not impact uric acid levels.
Various epidemiological studies have shown that purine-rich vegetables, whole grains, nuts and legumes, and less sugary fruits, coffee, and vitamin C supplements lower blood uric acid levels, but do not decrease the risk of gout. Red meat, fructose-containing beverages, and alcohol can increase the risk.
The role of uric acid in gout has been clearly defined and understood. As a result of this and the wide availability of relevant medications, gout is a very controllable form of arthritis.