Pneumologia
Asthme: un livre blanc présente 33 mesures pour améliorer la prise en charge

Paris, France — Alors que l’asthme tend à être banalisé, la pneumologie française a édité un livre blanc dans lequel sont exposées 33 propositions pour améliorer la prise en charge de cette pathologie, qui touche 3 à 5 millions de personnes en France [1]. Promouvoir l’éducation thérapeutique du patient, favoriser la pratique sportive, mais aussi lutter contre l’insalubrité font partie des mesures avancées.
Le document a été élaboré en partenariat avec les ministères des Sports et de l’Ecologie. Les principaux enjeux ont été présentés lors d’une conférence de presse, organisée en amont du 25ème congrès de pneumologie de langue française (CPLF), qui aura lieu sous forme virtuelle le 29 janvier [2].
L’objectif est notamment de sensibiliser sur les particularités de l’asthme sévère, qui affecte 5 à 10% des asthmatiques. « L’asthme sévère nécessite une prise en charge spécifique par un pneumologue de manière à avoir accès à des thérapies innovantes et ciblées », a indiqué le Pr Chantal Rahérison-Semjen (CHU de Bordeaux), présidente de la Société de pneumologie de langue française (SPLF).
Autres objectifs affichés, expliquant l’implication des deux ministères: trouver les moyens de « donner accès à une activité physique adaptée à tout patient asthmatique », notamment en milieu scolaire, et améliorer la qualité de l’air intérieur des logements, l’asthme étant considérée comme une maladie environnementale, a précisé la pneumologue.
Une pathologie marquée par les inégalités
L’appui politique s’est aussi traduit par l’engagement de la députée de Seine-Maritime, Agnès Firmin le Bodo, qui a déposé une proposition de loi dans le cadre de ce livre blanc pour lutter contre l’insalubrité liée à l’humidité. L’exposition aux moisissures, notamment chez les enfants, est considérée comme un facteur de risque d’asthme.
L’asthme reste une pathologie marquée par les inégalités. « Aujourd’hui, en fonction de l’endroit où l’on vit et des conditions environnementales, on ne peut pas avoir les mêmes chances d’évolution de la maladie asthmatique », a souligné le Pr Rahérison-Semjen, dans une vidéo présentant les principaux axes de proposition, publiée sur le site de la SPLF.
« Ces inégalités commencent dès l’accès aux soins : 40 % des patients suivis par leur médecin traitant n’auraient pas été orientés vers un médecin spécialiste en pneumologie ou en allergologie », notent les auteurs du document. Le recours aux urgences en cas de crise est aussi plus fréquent chez les patients plus précaires.
« Vingt ans après le plan asthme (2002-2005), peu d’avancées ont vu le jour pour améliorer le quotidien des patients et atténuer ces inégalités. Trop souvent, on considère à tort cette maladie comme banale et sans gravité ». Pourtant, en France, on compte chaque année 60 000 hospitalisations et 200 000 passages aux urgences pour asthme. Entre 800 et 900 patients en décèdent.
Six enjeux majeurs
Le livre blanc, destiné essentiellement aux pouvoirs publics et aux parlementaires, est le fruit d’un travail collaboratif entre la Fondation du souffle, la Fédération française de pneumologie et cinq sociétés savantes, dont la SPLF, ainsi que deux associations de patients. Ils ont formé le collectif « Asthme et inégalités » pour trouver des pistes d’amélioration.
Six enjeux majeurs ont été identifiés:
- Améliorer le parcours de soins du patient asthmatique;
- Renforcer la lutte contre le tabagisme des patients asthmatiques;
- Promouvoir l’éducation thérapeutique et la formation du patient;
- Favoriser la pratique sportive et l’activité physique des patients asthmatiques et lutter contre les idées reçues;
- Développer une culture et une connaissance de l’asthme au sein de la communauté éducative;
- Lutter contre les facteurs environnementaux.
Chacun de ces enjeux a été associé à plusieurs mesures pour un total de 33 propositions concrètes.
Promouvoir un examen du souffle
Concernant le parcours de soins, il est notamment suggéré de « systématiser l’orientation vers un pneumologue de tout patient passant aux urgences pour une crise d’asthme », « promouvoir la réalisation systématique d’un examen du souffle » ou « systématiser l’organisation de réunions de concertation pluridisciplinaire pour l’asthme sévère, afin de faciliter l’accès aux nouvelles thérapies ».
« Un des points noirs de la prise en charge des patients asthmatiques reste le suivi après un passage aux urgences. Beaucoup trop de patients n’intègrent pas un parcours de soins et nous savons qu’ils reviendront dans les mois qui suivent », précise dans le document le Pr Dominique Valeyre (hôpital Avicenne, AP-HP, Avicennes), président de la Fédération française de pneumologie.
A propos de la mesure de la fonction respiratoire, « trop peu de patients asthmatiques » y ont accès, précise le livre blanc. Or, elle permet « d’évaluer le contrôle de l’asthme, de montrer au patient la réalité de sa maladie et l’efficacité du traitement du souffle ».
Il est donc recommandé d’effectuer ce bilan régulièrement, « soit une fois par an pour les patients contrôlés, soit une fois tous les trois mois pour les patients non contrôlés ou souffrant d’un asthme sévère ». Ce type d’examen pourrait être réalisé au sein de structures locales, comme les maisons ou les centres de santé.
Des idées reçues sur la pratique sportive
L’accès à une activité physique adaptée représente également un enjeu majeur. « L’activité physique et la réhabilitation respiratoire sont des traitements non médicamenteux très importants dans la prise en charge des maladies respiratoires chroniques », a souligné le Pr Rahérison-Semjen, au cours de la conférence de presse.
Or, les idées reçues persistent. Selon la présidente de la SPLF, « un asthmatique doit pouvoir pratiquer le sport comme les autres, sans limitation quel que soit son niveau socio-économique. Malheureusement, nous entendons encore trop souvent aujourd’hui que les enfants asthmatiques ne peuvent pas faire de sport. »Un asthmatique doit pouvoir pratiquer le sport comme les autres, sans limitation quel que soit son niveau socio-économique. Pr Dominique Valeyre
Pour améliorer l’accès au sport, notamment en milieu scolaire, il est envisagé de mettre en place, en collaboration avec le ministère des Sports, une formation spécifique sur la pratique sportive chez les patients asthmatiques, mais aussi sur les fondamentaux concernant la maladie (traitement, prise en charge des crises…) pour « toute personne du milieu éducatif », a indiqué le Pr Rahérison-Semjen.
Former le personnel éducatif
« Nous allons poursuivre le travail pour que le personnel et la communauté éducative soit formé et accompagné, afin que les jeunes qui souffrent d’asthme puissent se voir proposer une activité physique adaptée et qu’ils ne se détournent pas de celle-ci de manière systématique », a affirmée Christèle Gautier, du ministère des Sports, lors d’une web-conférence de septembre dernier.
Le comité propose, par ailleurs, d’ « encourager les fédérations sportives à mettre en place un accueil adapté aux personnes asthmatiques », à l’image de DiabetAction, qui accompagne les patients diabétiques dans la pratique d’une activité physique. Il est aussi suggéré de développer un cours en ligne pour les encadrants sportifs, portant notamment sur la prévention des crises.
Autre proposition : « instaurer le remboursement de l’activité physique adaptée pour les patients asthmatiques les plus sévères ». Une mesure qui inciterait les médecins traitants à prescrire plus facilement une activité sportive.
Renforcer le recours au CMEI
Enfin, concernant l’aspect environnemental de la pathologie, le comité demande de « prendre des mesures concrètes, ambitieuses et efficaces contre la pollution pour réduire l’incidence de l’asthme et de l’asthme sévère ».
Il est notamment suggéré de soutenir les conseillers médicaux en environnement intérieur (CMEI), chargés d’enquêter au domicile du patient sur prescription médicale, afin de « mettre en œuvre des mesures pour l’éviction des allergènes (…) et des polluants domestiques », en vue d’améliorer l’habitat. Une intervention qui n’est pas encore intégrée dans la prise en charge.
Ce soutien peut être financier, avec un remboursement par l’Assurance maladie des visites à domicile prescrites par un médecin. Il est aussi proposé de doter ces spécialistes d’un statut « rendant obligatoire leur consultation ».
Des postes de CMEI avaient été promis lors du Grenelle de l’environnement de 2007, a précisé le Pr Rahérison-Semjen. « Malheureusement la promesse n’a pas été tenue. Nous a vraiment besoin de ces postes: avec un bon diagnostic de la qualité de l’air intérieur, on améliore le contrôle de l’asthme et la santé des patients. »
Actualités Medscape © 2021 WebMD, LLC
Citer cet article: Asthme: un livre blanc présente 33 mesures pour améliorer la prise en charge – Medscape – 27 janv 2021.
Ser mujer, fumar o haber fumado y D-dímero elevado, factores asociados a secuelas respiratorias post-covid
Conocer los factores que pueden predisponer a las secuelas pulmonares post-covid es esencial para identificar precozmente a los afectados a corto y medio plazo y establecer las medidas más adecuadas.

Conocer la magnitud, frecuencia y los factores de riesgo asociados a las secuelas respiratorias post-covid es un elemento clave, ya que poder identificar de forma temprana aquellos pacientes que con mayor probabilidad van a tener secuelas respiratorias tras superar la fase aguda de la covid-19 es determinante para optimizar los recursos técnicos y humanos en los laboratorios de función pulmonar y las consultas externas en un momento de máximo estrés del sistema sanitario.
Así lo indica un estudio desarrollado por el grupo de Infecciones Respiratorias del Instituto de Investigación Sanitaria del Hospital Universitario y Politécnico La Fe de Valencia (IIS-La Fe) en el propio centro sanitario, ha revelado que el sexo femenino, el tabaquismo (tanto fumadores actuales como exfumadores) o los D-dímeros elevados, son las variables clínicas que se relacionaron con una peor función pulmonar medida por DLCO (capacidad de difusión del pulmón para el monóxido de carbono (Diffusing capacity of the lung for carbon monoxide, por sus siglas en inglés). El estudio, publicado recientemente en Annals of the American Thoracic Society, ha sido dirigido por neumólogos y coordinado por Raúl Méndez y Rosario Menéndez, investigadora principal del grupo.
Según explica Méndez a Diario Médico, “la alta incidencia de casos ha saturado la asistencia clínica en atención primaria y a nivel hospitalario. Sin embargo, la necesidad de controlar y tratar las secuelas de los supervivientes está llevando también al colapso de los laboratorios de función pulmonar y las consultas externas de Neumología”.
Sin referencias conocidas
Por ello, y como punto de partida del estudio, se planteó el objetivo de evaluar las posibles secuelas respiratorias en los pacientes supervivientes de la covid-19 y, por extensión, las variantes clínicas asociadas a las mismas. “Al tratarse de una enfermedad nueva no existían referencias sobre las posibles secuelas respiratorias, la gravedad o la frecuencia de las mismas. La DLCO es una medida que se obtiene en el laboratorio de función pulmonar y que es capaz de identificar posibles secuelas o alteraciones en el intercambio gaseoso de los pulmones”, expone Méndez.
A partir de esa premisa, se realizó un estudio de seguimiento en la consulta externa de Neumología habilitada para el seguimiento de los pacientes con covid-19. “Se incluyó a todos los pacientes mayores de edad con diagnóstico confirmado de covid-19 que requirieron hospitalización en el Servicio de Neumología y/o la Unidad de Cuidados Intensivos (UCI). En total, se reclutaron 239 pacientes, de los cuales participaron 215. Ello lo convierte en el mayor estudio hasta la fecha que analiza las secuelas respiratorias de la covid-19 mediante el análisis de la DLCO”, comenta el investigador.
Datos en la escala de función pulmonar
Tras obtener los resultados de esta prueba respiratoria se analizaron los datos de función pulmonar que presentaban. “Entre los principales resultados cabe destacar que un 24,7 por ciento de los pacientes tenían una alteración de la DLCO (se considera alteración tener una DLCO <80% de acuerdo a la población normal según edad, peso, sexo y estatura). De los pacientes con una DLCO alterada, un 75,5 por ciento tenían una alteración leve (DLCO entre el 80 y el 60%), un 24,5 por ciento una alteración moderada (DLCO entre el 60 y el 40%) y ninguno tuvo alteración grave (DLCO <40%)”.
Para Méndez, estos resultados “son preocupantes y esperanzadores a la vez: preocupantes porque casi un 25 por ciento con una incidencia acumulada tan alta supone un gran volumen de pacientes con secuelas; esperanzadores porque probablemente los neumólogos esperábamos un mayor porcentaje de pacientes con secuelas respiratorias y de una mayor gravedad”. De todas formas, ha advertido, hay que tomar estos datos con cautela ya que el estudio ‘solo’ incluye 40 pacientes que requirieron ingreso en UCI. “Se necesitan más estudios y de mayor tamaño para analizar por ejemplo con más detalle las secuelas específicamente en los pacientes de UCI. Además, se desconocen si estas secuelas van a persistir o a remitir a largo plazo”, apunta.
Además del impacto global, el grupo analizó y comprobó que las variables clínicas que se relacionaron con una peor función pulmonar medida por DLCO fue el ser mujer, tabaquismo (tanto fumadores actuales como exfumadores), D-dímeros elevados (los D-dímeros son un producto de degradación de la fibrina y que se relaciona con problemas vasculares y tromboembólicos) y, aunque sin alcanzar significación estadística pero sí una clara tendencia, haber requerido hospitalización en la Unidad de Cuidados Intensivos (si bien este grupo era pequeño).
Factores analizados al detalle
Estos resultados, según Méndez, tienen distintas explicaciones debido a las características propias de la covid-19. “El tabaquismo y las enfermedades respiratorias crónicas que provoca se relacionan en ocasiones con alteraciones de la DLCO. Este hallazgo, por tanto, puede reflejar la presencia de un daño pulmonar previo o incluso agravado por la covid-19”, comenta.
En relación a los D-dímeros, cabe destacar que la infección por SARS-CoV-2 se ha relacionado estrechamente en diversos estudios con problemas vasculares y trombóticos. “Estos problemas vasculares a nivel pulmonar en ocasiones pueden dejar como secuela una alteración del intercambio gaseoso con un deterioro de la DLCO”, expone.
En tercer lugar, las mujeres tienen más riesgo de tener secuelas en la DLCO. “Si bien durante la fase aguda son los hombres los que presentan un cuadro más grave, diversos estudios muestran que las secuelas o síntomas persistentes en forma de lo que se ha llamado long-COVID es más prevalente en las mujeres”, remarca el especialista. Por último, los pacientes que requirieron ingreso en UCI mostraron también una tendencia a tener peor DLCO: “Estos pacientes han presentado una mayor gravedad y es esperable un mayor impacto a nivel respiratorio tras la fase aguda”.
El trabajo de este grupo ha puesto el foco sobre el impacto y las variables clínicas asociadas al riesgo de padecer alteraciones respiratorias post-covid. Pero el nuevo reto a corto y medio plazo es saber su posible extensión o gravedad.
Según señala Méndez, uno de los coordinadores del estudio, “los objetivos serían conocer a más largo plazo si la alteración de la DLCO es permanente o mejora a lo largo del tiempo (6 ó 12 meses) y los factores asociados a esta posible evolución”. Dado el impacto de las patologías respiratorias en la población general, el especialista considera que “sería interesante ampliar si es posible la población a estudio con más pacientes procedentes de la Unidad de Cuidados Intensivos (UCI) y con daño previo de la función pulmonar, como por ejemplo pacientes con enfermedades respiratorias crónicas (asma o EPOC, entre otras)”.
El grupo de Infecciones Respiratorias del IIS-La Fe ha multiplicado su actividad investigadora desde que empezó la pandemia, redirigiendo gran parte del esfuerzo a investigar acerca de la covid-19. Además del presente estudio, están desarrollando distintas líneas de investigación frente al SARS-CoV-2. “Destacan por ejemplo el estudio de las secuelas cognitivas, psiquiátricas y sobre la calidad de vida a corto y largo plazo. Este tema preocupa a los clínicos por los hallazgos encontrados en el seguimiento de los supervivientes. Este trabajo, realizado junto con psiquiatras, acaba de ser publicado en Journal of Internal Medicine y aporta información valiosa en este campo”, comenta Méndez.
Avance de estrategias
El grupo de investigación tiene además una amplia experiencia en el análisis de biomarcadores pronósticos en neumonía adquirida en la comunidad (no covid). En la actualidad tienen varios trabajos en revisión sobre marcadores pronósticos que analizan entre otras cosas el daño endotelial agudo y residual provocado por el virus o el riesgo trombótico asociado.
Algunos de estos trabajos se encuentran actualmente en revisión para su publicación en revistas científicas. Además, junto con estas y otras líneas de investigación propias, el grupo de Infecciones Respiratorias, con Rosario Menéndez al frente, coordina varios estudios multicéntricos a nivel nacional y colabora en otros como el proyecto CiberRES.
Sugieren que el contagio de coronavirus por aerosoles o por vía intranasal origina una enfermedad más grave
Nuevos estudios preliminares relacionan la gravedad de la covid-19 con la vía de transmisión, por lo que se recomienda seguir con las medidas de ventilación adecuada.
La transmisión de enfermedades por vía aérea es algo establecido para muchas enfermedades, fundamentalmente las respiratorias como el coronavirus. Las vías clásicas de contagio en esta pandemia son por fómites al tocar superficies, por gotas expelidas por una persona portadora del virus y la tercera, que algunos especialistas consideran la más relevante, es la que se propaga a través del aire por aerosoles, “pequeñas gotículas de saliva y de fluidos respiratorios más pequeñas –de menos de 50 micras, que se quedan suspendidas en el aire, entre 1 y 2 horas, y con capacidad infectiva cuando las respiramos, puesto que el virus mide 0,1 micras. “Son muy peligrosas, sobre todo, cuando se producen en interiores, en zonas sin ventilación y aunque se mantenga la distancia”, señalada a DM José Luis Jiménez, catedrático de Química y Ciencias Ambientales de la Universidad de Colorado, Boulder, (Estados Unidos) y uno de los mayores expertos mundiales en aerosoles que, desde que empezó la pandemia, y junto a otros especialistas, insiste en la transmisión del SARS-Cov-2 a través de aerosoles como vía dominante.
Fuente de contagio y de posible gravedad
A pesar de que la contribución de cada una de las vías de transmisión se sigue analizando y de que la Organización Mundial de la Salud (OMS) no se ha pronunciado tajantemente sobre este aspecto -ha hecho ‘pequeños guiños’ sobre su peso específico- cada vez más son las instituciones y administraciones sanitarias y estudios científicos que confirman la transmisión del SARS-CoV-2 a través de aerosoles. Jiménez, de hecho, la considera “responsable directa de la propagación imparable de la pandemia”, señalando que hasta el 75% de los contagios se produce por esta vía, por lo que la necesidad de ventilar espacios interiores es esencial.
Si a los primeros datos que publicó Science se fueron sumando los de otras publicaciones científicas, nuevos datos de un ‘preprint’, estudio no revisado por pares, de los Institutos Nacionales de Salud (NIH) de Estados Unidos, que aparece en bioRxiv, sugiere que la gravedad del Covid-19 depende de la ruta de infección, según los resultados preliminares realizados en modelo experimental. Así, sugieren que la infección por inhalación de aerosoles o vía intranasal provocaría una enfermedad más grave que la infección por fómites.
Los ensayos se han llevado a cabo con hámsteres sirios a los que infectaron con el SARS-CoV-2 por diferentes vías: intranasal, aerosol y fómites. La intranasal y la que se propaga por aerosoles causaron patologías respiratorias más graves y tenían mayores cargas virales, frente al contagio que se observó con la exposición a fómites, que originaba una enfermedad más leve caracterizada por un estado inmunológico antiinflamatorio y un patrón de eliminación retardado, según Vincent J. Munster, del Laboratorio de Virología de los NIH estadounidenses, y miembro del equipo que ha realizado este análisis comparativo.
En comparación con la exposición intranasal, los animales expuestos a aerosoles tenían una carga viral más alta en la tráquea y el pulmón, lo que sugiere que el aerosol deposita núcleos de gotitas virales de SARS-CoV-2 en el sistema respiratorio inferior, con una replicación más rápida en el pulmón, más eficientemente, aunque este tipo de diseminación también se observa en el tracto respiratorio superior.
Invertir el flujo de aire
Sin embargo, y según Julia R. Port, del mismo Laboratorio de Virología, cuando se invertía el flujo de aire de los animales no infectados hacia los animales infectados, se observaba una fuerte reducción en la transmisión. “El flujo de aire direccional juega un papel importante en la transmisión del SARS-CoV-2”, hecho que ya se observado en espacios cerrados y mal ventilados, por lo que insisten en elevar las medidas preventivas basadas en la ventilación de interiores que ayudarán directamente al control de la pandemia“.
A pesar de que aún no hay datos concluyentes en cuanto al peso específico de la vía de contagio de la infección en la gravedad de la covid-19 en humanos, sí parece que “los aerosoles más grandes se depositan en la parte superior de la garganta, nariz y región traqueobronquial del pulmón y que los aerosoles realmente pequeños, los de menos de 1 micra, pueden penetrar hasta los alveolos pulmonares”, subraya Munster.
En espera de la confirmación de la relación directa entre gravedad de la infección y la vía de contagio, los investigadores piden ahondar en las medidas encaminadas a ventilar espacios cerrados y a medir las concentraciones de CO2 todo lo que sea posible, como una forma de reducir las posibilidades de contagio.
“Ventilar, ventilar, ventilar espacios cerrados”, era el mensaje clave de José Luis Jiménez. Si no se puede, “medir el CO2 del aire o recurrir a filtradores HEPA. Las medidas para evitar esta transmisión deben explicarse y conocerse sin causar alarma ni asustar a la población, pero deben tenerse en cuenta y poner el máximo esfuerzo en interiores, que es donde se están produciendo la gran mayoría de los contagios”, señala este experto mundial.
Problemas neurológicos, psicológicos o motores, entre los síntomas de la covid persistente
Mujer de 43 años con síntomas durante más 185 días es el perfil más frecuente del afectado por covid persistente, según un sondeo a 2.120 personas.

El perfil más frecuente del enfermos de covid persistente es de una mujer de 43 años, que lleva más de 185 días con síntomas persistentes de covid-19 tras haber sido contagiada de la enfermedad en la primera ola de la pandemia. Además de los síntomas más generales asociados a la infección, también presenta otros neurológicos, psicológicos, motores o respiratorios.
Es el ‘retrato robot’ ofrecido por la encuesta lanzada por la Sociedad Española de Médicos Generales y de Familia (SEMG) y los colectivos de afectados Long Covid ACTS entre el 13 de julio y el 14 de octubre de este año.
Los médicos piden profundizar en las causas para tratar adecuadamente estos casos y además minimizar los riesgos de quien sea más susceptible de sufrir indefinidamente las consecuencias de esta enfermedad. Antonio Fernández-Pro, presidente de la SEMG, ha señalado que los médicos dieron la voz de alarma cuando observaron la persistencia de los síntomas en un gran número de pacientes, “pero hay muchas preguntas que nos quedan por contestar, y la respuesta que podemos dar hoy en noviembre de 2020 puede ser distinta en marzo de 2021”.
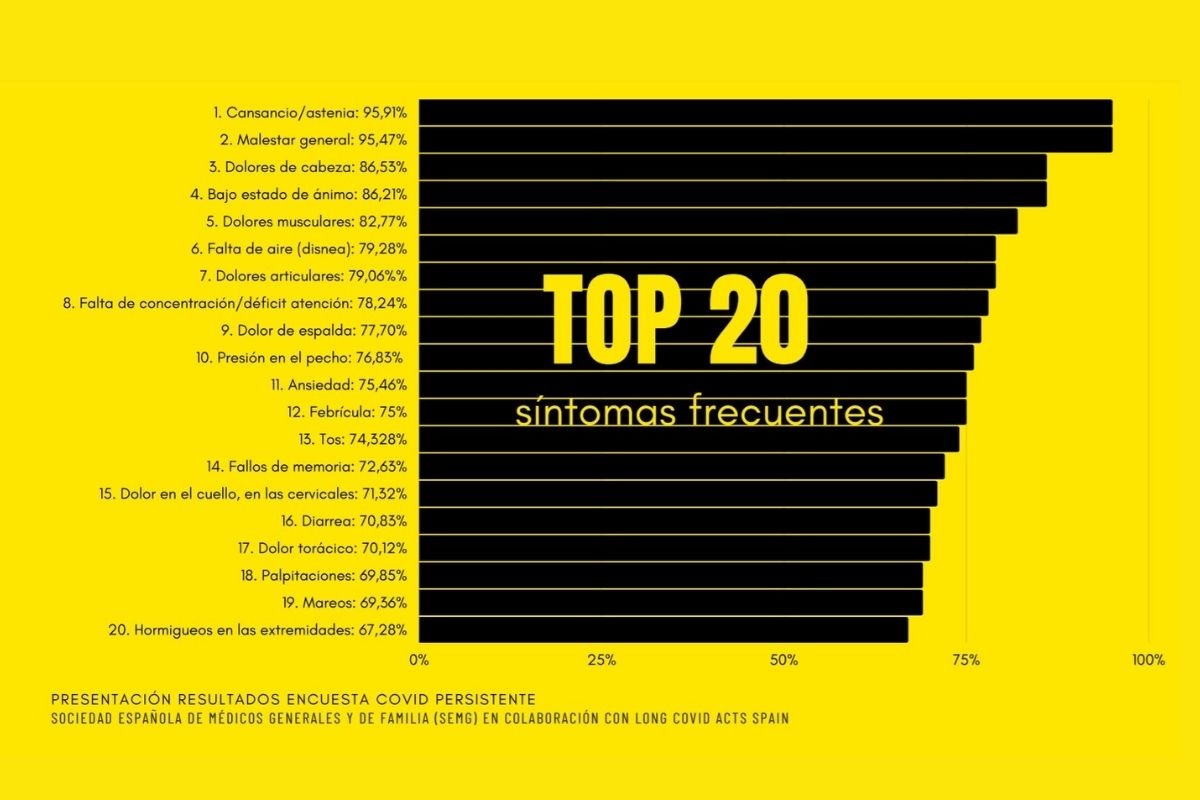
Durante esos tres meses, el sondeo fue contestado por un total de 2.120 personas, de las cuales, 1.834 son enfermos que presentan síntomas compatibles de covid persistente o de larga duración. La mitad de los enfermos tenían edades comprendidas entre los 36 y los 50 años de edad y procedían de las comunidades de Madrid (35%) y Cataluña (30%), que fueron las más castigadas al principio de la pandemia.
En la encuesta sobre covid persistente se han registrado hasta un total de 200 síntomas persistentes que fluctúan y se presentan en el tiempo, con una media de 36 síntomas por persona. Los más frecuentes son cansancio/astenia; malestar general; dolores de cabeza; bajo estado de ánimo; dolores musculares o mialgias; disnea; dolores articulares y falta de concentración y déficit atención, entre otros.
Pilar Rodríguez, vicepresidenta 1º de la SEMG, ha apuntado que, si bien tras muchas infecciones se producen síndromes post virales, “en lo que no se parece a otros casos es que este síndrome dura meses y meses y además la afectación es multiorgánica“.
El 50% de los encuestados tiene 7 áreas afectadas, siendo las más frecuentes los síntomas generales (95%), las alteraciones neurológicas (86%), los problemas psicológicos/emocionales (86%), los problemas del aparato locomotor (82%), respiratorios (79%), las alteraciones digestivas (70%), cardiovasculares (69%), otorrinolaringológicas (65%), oftalmológicas (56%), dermatológicas (56%), alteraciones de la coagulación (38%) y las alteraciones nefrourológicas (25%).
Hay que señalar que al 78% de los encuestados se les hizo prueba diagnóstica, siendo las más frecuentes la PCR, que constató en un 73% resultado positivo. Del mismo modo, de la encuesta se desprende que al 59% del total se le hizo prueba de seguimiento.
A pesar de los problemas de acceso a pruebas durante la primera ola de la covid-19, la encuesta no muestra diferencias estadísticamente significativas entre aquellos que tuvieron acceso a las pruebas diagnósticas y los que no, ni tampoco entre los que los resultados fueron positivos o ya no lo fueron, probablemente porque se hicieron tarde.
Síntomas incapacitantes
Los pacientes tienen una mala percepción subjetiva de su estado de salud y su incapacidad actual, con ambos valores puntuados entre 0-5 en una puntuación sobre 10 por el 50% de los encuestados.
La vicepresidenta 1º de la SEMG ha destacado que los síntomas más prevalentes (cansando, dolor de cabeza) son los más incapacitantes. “La característica común, aparte de la edad y el género, es que son personas sanas, y quizás es una idea que tengamos que transmitir a los jóvenes. Es una afectación que va más allá de la salud: es un problema social, familiar, laboral y económico“.
En su vida diaria, el 30,43% de los encuestados afirma que le supone un esfuerzo o les resulta imposible algo tan básico como el aseo personal; al 67,99% la realización de las actividades de casa; al 72,52% trabajar fuera de casa; el 70,12% tienen dificultades para atender a las obligaciones familiares diarias y al 74,65% le supone un esfuerzo o imposible el ocio con los amigos y otras personas.
Anna Kemp, portavoz del colectivo de enfermos, pedía reconocimiento oficial a la figura del paciente de covid persistente y que empiece a figurar también las estadísticas: “Nos enfrentamos cada día a una enfermedad desconocida, no reconocida y muy incapacitante. Da miedo y tiene mucho impacto, pierdes todo lo que te define y sostiene. En sociedad nos vemos a veces solos y bastante aislados, muchos tienen problemas de falta de comprensión por parte de sus jefes y compañeros de trabajo”.
En el ámbito sanitario “nos sentimos bastante incomprendidos, frustrados y no acompañados. Necesitamos ese reconocimiento, estamos hartos de tener que demostrar que estamos enfermos, llevamos 6-7 meses así”.
“Necesitamos un cambio de paradigma; se habla de secuelas tras haber superado covid y no está nada claro que sea así”, ha señalado Silvia Guerrero, otra de las portavoces del colectivo de pacientes.
Lorenzo Armenteros, portavoz covid-19 de la SEMG, destacó que una dificultad añadida en estos casos es que “las pruebas que se realizan a estos pacientes dan valores normales, pero eso no significa que no tengan nada. Necesitamos unas pruebas y unas escalas específicas; por ejemplo, buscar el virus en otras parte del cuerpo, como el tracto digestivo”.
Líneas de investigación
Rodríguez ha apuntado que es necesario profundizar en la investigación, con la vista puesta más allá de dar tratamiento a los casos más graves. “El primer objetivo es salvar la vida de la gente, pero en el futuro tendremos como poco dos problemas: las personas en peligro vital y las que están vitalmente afectadas durante meses. Hay que intentar disminuir los síntomas o incluso curar a quienes tienen covid persistente, revertir su situación para que puedan hacer una vida normal”.
Rodríguez ha adelantado que la SEMG empezará próximamente un ensayo para identificar las causas y detectar tratamientos específicos. Además, va a poner un marcha un registro clínico de pacientes afectados por covid, independientemente de su gravedad. El registro tiene dos objetivos: caracterizar bien a los pacientes y hacer seguimiento a los que tengan mayor problemática y “estratificar a la población para saber quiénes tienen más riesgo de desarrollar una covid persistente, porque se podrían de algún tratamiento precoz y así minimizar los riesgos”.
Covid-19 e doença de Chagas: Alerta vermelho

No dia 22 de maio, um homem de 69 anos foi hospitalizado na área covid-19 do Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (HCFMUSP). O paciente, que fazia acompanhamento para doença de Chagas sem patologia comprovada, apresentava problemas respiratórios. Uma semana depois, uma mulher de 74 anos com cardiomiopatia crônica por doença de Chagas foi internada com sintomas associados a covid-19. Após uma rápida progressão da doença, os dois morreram.
“Acreditamos que a doença de Chagas possa ser um importante e subestimado fator de risco de covid-19 grave, especialmente para aqueles com doença de Chagas crônica e cardiomiopatia, que podem apresentar maior probabilidade de desfechos ruins”, alertaram os autores do que é, talvez, um dos primeiros relatos de caso de pacientes com doença de Chagas e covid-19 concomitantes. [1]
O alerta é para as áreas endêmicas com infecção por doença de Chagas subnotificada e/ou subnotificação da infecção por SARS-CoV-2 – situação que abarca toda a América Latina.
Segundo o epidemiologista Dr. Sergio Sosa-Estani, Ph.D., diretor do Programa Clínico de Chagas da iniciativa Medicamentos para Doenças Negligenciadas (DNDi, sigla do inglês, Drugs for Neglected Diseases initiative), os portadores de Trypanosoma cruzi já não residem apenas nas regiões rurais, eles também “estão nas cidades grandes, às vezes porque migraram, outras porque são filhos ou netos de mãe infectada, ou porque foram contaminados com o parasita por transfusão sanguínea antes dos protocolos de controle do sangue, que começaram no final dos anos 70”.
As comorbidades tornam os dois grupos de pacientes vulneráveis. Assim como em pacientes com covid-19 grave, vários estudos descrevem altos níveis de comorbidades em pacientes com doença de Chagas. Um estudo brasileiro identificou uma média de 2,7 comorbidades crônicas em pacientes com doença de Chagas. [2] Dentre 168 pacientes com doença de Chagas em São Paulo, 51,2% tinham hipertensão e 23,8% tinham diabetes. [3] Essas comorbidades também refletem a idade mais avançada das populações que são especialmente impactadas pela doença de Chagas e pela covid-19.
Alerta amarelo ou vermelho
Se no momento da admissão hospitalar o paciente com covid-19 tiver infecção por T cruzi. sem patologia comprovada, deve-se acender uma luz amarela, mas caso ele tenha doença de Chagas com patologia confirmada, essa luz deve ser vermelha, resumiu o Dr. Sergio a pedido do Medscape.
O Dr. Álvaro Avezum, cardiologista e diretor do Centro Internacional de Pesquisa do Hospital Alemão Oswaldo Cruz, em São Paulo, que conduz ensaios clínicos sobre covid-19 e doença de Chagas, concorda: “Muita atenção nessa hora. Qualquer paciente com doença cardiovascular é de risco maior quando contrai covid-19. Se o comprometimento cardíaco ocorrer por conta da doença de Chagas, teremos duas situações; a questão cardiovascular e a doença imune da doença de Chagas.”
A covid-19 tem sido associada a múltiplas manifestações cardíacas, que incluem arritmias, infarto do miocárdio, exacerbações de insuficiência cardíaca e miocardite fulminante. Possíveis complicações entre covid-19 e cardiopatia chagásica podem ser esperadas, principalmente devido às vias imunológicas comuns compartilhadas por ambas as doenças, como a enzima conversora de angiotensina 2 (ECA2), que está envolvida na função cardíaca e no desenvolvimento de hipertensão arterial e diabetes, fatores de risco frequentemente observados em pacientes com cardiopatia chagásica. Entre as questões a serem estudadas está a ligação entre a inflamação causada pela covid-19 e a causada pela doença de Chagas.
Indivíduos com doença de Chagas podem ter progredido para uma forma cardíaca da doença sem saber. “O acometimento cardíaco da doença de Chagas ocorre em pessoas jovens, que podem ser absolutamente saudáveis e não ter risco de cardiopatia por T. cruzi. Há casos de morte súbita de pessoas jovens com T. cruzi sem sintomatologia prévia. A nossa preocupação agora é como pode seguir a coinfecção? Poderia acelerar o processo de manifestação cardíaca? Seria desencadeante de um desenlace fatal?” indagou o Dr. Sergio.
“Se um paciente tem alterações de coagulação causadas pela covid-19 e tem cardiopatia chagásica inicial, o risco de tromboembolia pode estar aumentado. Teme-se também que a tormenta imunológica possa ativar um parasita que até então estava em equilibro com seu hospedeiro e alterar o curso da doença.”
Outro aspecto preocupante são as possíveis sequelas da covid-19; “não apenas as cardíacas, mas a fibrose pulmonar é capaz de produzir um grau de resistência à circulação pulmonar que pode causar sobrecarga no coração e complicar a evolução de uma cardiopatia chagásica”, acrescentou o Dr. Sergio.
O Dr. Sergio liderou um grupo de especialistas em doença de Chagas e doenças cardíacas que avaliou as informações disponíveis e chegou a um consenso, que acaba de ser publicado no periódico Global Heart.[3] Foram estudados, entre outras questões, os dados sobre o uso de cloroquina, hidroxicloroquina, inibidores de protease (ritonavir, liponavir), azitromicina, atazanavir, ivermectina, entre outros. Diante do comprometimento cardíaco causado pela covid-19, somado ao causado pela doença de Chagas, “o que se faz é tratar as duas condições, sabendo que podem haver interações medicamentosas. Mas ainda temos poucas evidências científicas. Por isso o artigo é útil, porque traz um guia, uma recomendação com o estado atual do nosso conhecimento”, avaliou o Dr. Álvaro, que não participou da redação do artigo.
Pacientes com as duas doenças podem receber terapia imunossupressora, mas recomenda-se um acompanhamento rigoroso para diagnosticar a reativação da doença de Chagas no início do curso. No caso de evidência clínica e/ou parasitológica de reativação, deve-se iniciar o tratamento com benzonidazol ou nifurtimox. Em caso de uso de benzonidazol e nifurtimox em combinação com medicamentos para tratar a covid-19, a hepatotoxicidade e a função hematológica devem ser monitoradas.
Podem ocorrer interações medicamentosas em pacientes com cardiopatia chagásica sendo tratados para arritmias cardíacas, como fibrilação atrial ou arritmias ventriculares, com risco de vida que recebem amiodarona, em função do potencial aumento do intervalo de QT associado aos tratamentos propostos para a covid-19.
O Dr. Sergio acredita que realizar a sorologia para T. cruzi no momento da admissão hospitalar de pacientes com covid-19 pode ajudar a orientar o atendimento.
“Se o paciente tem cardiopatia, é importante saber a causa. Se não tiver cardiopatia, é bom ter o conhecimento, porque não sabemos se o parasita pode se reativar de forma grave.”
Pacientes com doença de Chagas na pandemia
Dependendo da situação epidemiológica, os médicos precisam ponderar os riscos e os benefícios de encaminhar um paciente com doença de Chagas em fase crônica indeterminada (o que pode durar anos ou décadas) para um centro de saúde. O médico precisa saber quando iniciar ou postergar o início de um tratamento, ou prescrever um fármaco sobre o qual tem dúvida quanto a potenciais interações medicamentosas.
Na opinião do grupo de trabalho, formas agudas da doença de Chagas geralmente justificam tratamento antiparasitário o mais cedo possível, mesmo no contexto da pandemia. O tratamento antiparasitário continua sendo necessário se, além da doença de Chagas, o paciente tiver covid-19 – mas os médicos devem ficar atentos à gravidade dos sintomas da infecção causada pelo SARS-CoV-2.
No caso da transmissão congênita, a criança infectada, mas sem sintomas de covid-19, deve receber tratamento antiparasitário assim que o diagnóstico da infecção por T. cruzi for estabelecido.
Em outros casos, o tratamento antiparasitário pode ser adiado até que o paciente possa frequentar o hospital com segurança, no entanto, a reativação da doença de Chagas com sinais em órgãos-alvo deve ser acompanhada de perto. Os pacientes em reativação devem ser internados e receber tratamento antiparasitário por 60 dias, anti-histamínicos e/ou anti-inflamatórios. A reativação envolvendo miocardite/meningoencefalite é de particular preocupação, devendo ser cuidadosamente monitorada na unidade de terapia intensiva.
Se um paciente já estiver recebendo benzonidazol ou nifurtimox, o tratamento deve ser continuado com as devidas medidas de isolamento. Os pacientes com cardiopatia chagásica devem continuar seus tratamentos habituais durante a pandemia. Não há nenhuma evidência até o momento para apoiar a descontinuação de inibidores da ECA e/ou bloqueadores de receptores da angiotensina (BRA) com base na hipótese de aumento da suscetibilidade à infecção pelo SARS-CoV-2.
As indicações para triagem e diagnóstico de doença de Chagas não foram alteradas, mas a urgência depende do grau em que o diagnóstico da doença de Chagas afetará a gestão de curto prazo do indivíduo. Gestantes, bebês nascidos de mães soropositivas, pacientes com síndromes clínicas sugestivas de doença de Chagas e qualquer indivíduo que receberá iminentemente imunossupressão deve ser testados para orientar avaliação e terapia.
O uso de ferramentas para consulta virtual é altamente recomendado. Para os pacientes com doença de Chagas, a ida às unidades de saúde seria indicada principalmente para a realização de exames, priorizando a realização das consultas por meio de telemedicina. A equipe de saúde do Instituto Nacional de Infectologia Evandro Chagas, centro nacional de referência para diagnóstico e tratamento de doenças infecciosas, que atende regularmente mais de 1.000 pacientes com doença de Chagas crônica sob tratamento integral, iniciou atendimento telefônico para todos os pacientes com cardiomiopatia chagásica crônica inscritos no programa de reabilitação cardiovascular antes da pandemia covid-19.
A equipe estabeleceu um questionário e organizou uma escala de serviço para contatar os pacientes. Além de fornecer orientações gerais sobre cuidados de saúde durante a pandemia, a equipe preenche um questionário que permite coletar informações gerais e detectar mudanças significativas e presença de sintomas.
“Acreditamos que essa estratégia de suporte telefônico pode ajudar, pelo menos em parte, na manutenção da assistência integral aos nossos pacientes, e pode nos alertar para sinais precoces e sintomas de descompensação, proporcionando a oportunidade de intervenção antes que os pacientes precisem de internação.” [5]
Incertezas
A compreensão das possíveis relações entre a doença de Chagas e a covid-19 ainda é limitada. Há muitas lacunas no conhecimento sobre os cuidados em relação ao tratamento da covid-19 em pacientes com doença de Chagas, riscos hemodinâmicos e eletrofisiológicos da covid-19 em pacientes com cardiopatia chagásica, efeitos antivirais de antiparasitários, influência dos anti-inflamatórios e impacto da terapia anticoagulante nas duas doenças. Tratamentos cardiovasculares de doença de Chagas, como amiodarona, podem tratar a covid-19? A tempestade de citocina pode desencadear reativação parasitaria ou progressão da doença? Há reação cruzada entre as vias de resposta imune, viral e parasitária? O estado pró-trombótico de ambas as doenças se comporta sinergicamente? O estado inflamatório crônico da doença de Chagas agrava o quadro de covid-19?
Em resumo, ainda não se sabe como a história natural das duas doenças é afetada pela coinfecção, “a priorização da atenção à pandemia de covid-19 não deve tirar a atenção de outros problemas que afetam a nossa população, especialmente as doenças negligenciadas, das quais a de Chagas é a mais relevante na região”, concluiu o Dr. Sergio.
Os Drs. Sergio Sosa-Estani e Álvaro Avezum informaram não ter conflitos de interesses.
Medscape Notícias Médicas © 2020 WebMD, LLC
Citar este artigo: Covid-19 e doença de Chagas: Alerta vermelho – Medscape – 25 de novembro de 2020.
La EPOC triplica la mortalidad por covid-19
En el Día Mundial de la EPOC, que se conmemora hoy, los neumólogos alertan de los mayores riesgos de estos pacientes.
La tasa de mortalidad para los pacientes mayores de 40 años es casi el triple respecto a la población general cuando el hospitalizado por coronavirus padece además enfermedad pulmonar obstructiva crónica (EPOC), según los resultados de un estudio dirigido por investigadores españoles a partir de miles de historias clínicas analizadas con inteligencia artificial y procesamiento del lenguaje natural.
La investigación coordinada por José Luis Izquierdo, del Hospital de Guadalajara, y Joan B. Soriano y Julio Ancochea, del Hospital La Princesa de Madrid, ha contado con el soporte de Savana, la plataforma de inteligencia artificial que acelera y expande la investigación médica. Asimismo, los pacientes con EPOC tienen mayor propensión a cardiopatía isquémica y otros factores que les ponen en riesgo por complicaciones asociadas a la covid-19.
El estudio, titulado ‘Características y pronóstico de la covid-19 en pacientes con EPOC’, se publica en Journal of Clinical Medicine y forma parte del estudio internacional Big COVIData que puso en marcha Savana al inicio de la pandemia. Revela que el perfil del enfermo con EPOC más propenso a la infección es varón -en cuatro de cada cinco casos-, con 75 años de edad y otras patologías añadidas (diabetes, arritmias e insuficiencia cardíaca, entre otras).
Para el estudio, se partió de una muestra de datos de 1,16 millones de pacientes en Castilla-La Mancha, posteriormente filtrados y analizados de forma anonimizada entre el 1 de enero y el 10 de mayo con la tecnología EHRead de Savana, implantada ya en una decena de países en Europa y Norteamérica, que utiliza machine learning (aprendizaje automático) y procesamiento del lenguaje natural para la lectura de las anotaciones que miles de profesionales sanitarios recogen cada día en las historias clínicas.
Hospitalización elevada
“Una de las principales preocupaciones que tuvimos cuando aparecieron los primeros casos de covid-19 era conocer cuál iba a ser su impacto en las patologías respiratorias crónicas. Pronto supimos que la gravedad era mayor en hipertensos, diabéticos o con enfermedad cardiovascular. Este estudio supone una de las series más amplias de la literatura médica sobre el impacto de la covid-19 en pacientes con EPOC utilizando Big data e inteligencia artificial”, explica José Luis Izquierdo Alonso, jefe de Neumología del Hospital de Guadalajara.
Del total de pacientes involucrados en el estudio, algo más de 31.600 padecían EPOC y de ellos 793 resultaron contagiados por el coronavirus. En población sin EPOC y con covid-19, la cifra se situó en 13.546. De los resultados se desprende que los enfermos de EPOC muestran tasas más altas de diagnóstico de la covid-19 (2,51%) que la población mayor de 40 años sin patología respiratoria (1,16%).
Sus tasas de hospitalización se sitúan en el 40% frente a poco más del 31% y los porcentajes de mortalidad de quienes ingresan en el hospital por covid-19 casi se triplican cuando hay EPOC: 9,3% frente al 3,4 %. En cuanto al perfil del paciente con EPOC más propenso de contagiarse, los datos desvelan que se trata de un varón en el 83% de los casos, con una media de edad de 75 años frente a los 66 de la población general con coronavirus. Muestran mayor prevalencia de comorbilidad o enfermedades añadidas que quienes solo tienen coronavirus, y fuman con mayor asiduidad. La neumonía fue el diagnóstico más común entre los pacientes con EPOC hospitalizados a causa del coronavirus (59 %). El 19% presentaba infiltraciones pulmonares sugestivas de neumonía e insuficiencia cardíaca.
“La principal conclusión -resume Izquierdo- es que los pacientes con EPOC tienen el doble de riesgo de contraer la enfermedad y cuando esto ocurre tres veces más posibilidades de fallecer. Esto no debe servir para generar angustia sino para extremar las medidas de autoprotección, especialmente con los contactos, pero sin prescindir de una vida activa, ya que mantener una actividad física regular es muy importante para el control de la enfermedad”.
Controversia sobre inhaladores
Este estudio coincide con una revisión de quince estudios publicada en European Respiratory Review por un equipo de la Universidad británica de Manchester que corrobora que tener EPOC supone un mayor riesgo de contagio de la covid-19, así como peores resultados clínicos, incluida una tasa de mayor mortalidad.
Según ese análisis, entre los mecanismos que aumentan la susceptibilidad a la infección de SARS-CoV-2 en el caso de pacientes con EPOC figura el aumento de la expresión de ECA2 pulmonar, el receptor que permite la entrada del SARS-CoV-2 en las células y que, por lo tanto, hace más susceptibles a estos pacientes. A este se suma que tienen una defensa antiviral reducida, células endoteliales disfuncionales y aumento de la coagulopatía, que pueden empeorar el curso clínico de la covid-19.
Con respecto al debate sobre si el uso de los corticosteroides inhalados, empleados para el control de la EPOC, podrían protegerles de la covid-19, la revisión no ha obtenido una respuesta clara: “No hay evidencia de que protejan frente a la covid-19 ni de que estén asociados a peores resultados clínicos”. Por un lado, reducen la replicación del SARS-CoV-2 y la expresión de ECA2, mediante la disminución de la producción de interferón 1, pero, por otra parte, este descenso de interferón 1 se ha asociado a que tengan afectadas las defensas, en particular las células T.
En cuanto a los resultados clínicos de la covid-19 en pacientes con EPOC, la revisión de estudios de cohortes no ha demostrado que sean más o menos susceptibles de contraer la infección, pero sí que tienen un mayor riesgo de sufrir peores resultados, que incluyen la necesidad de ventilación mecánica, y mayor mortalidad, si bien dada la diversidad de estudios analizados, las tasas de fatalidad varían de forma muy llamativa entre el 1% y el 62%.
Aun así, un metanálisis de 11 series de casos de China y Estados Unidos halló un aumento del riesgo del 88% entre los pacientes con EPOC infectados de requerir un ingreso en UCI o de morir por covid-19. El estudio con la mayor cohorte, de 13.442 pacientes atendidos en urgencias en Nueva York, encontró un riesgo aumentado de hospitalización del 1,77 y una tendencia hacia una mayor mortalidad, cifrada en un riesgo relativo de 1,08.
Otro estudio italiano, de una cohorte de 1.044 pacientes hospitalizados con EPOC, detectó que tenían un riesgo aumentado y significativo de fracaso respiratorio grave, cifrado en un riesgo relativo de 1,17. Y en España, un estudio de una cohorte longitudinal halló que presentan un 70% más riesgo de fallecer.
Según José Luis López Campos, neumólogo y coordinador del Área EPOC de la Sociedad Española de Neumología y Cirugía Torácica (Separ), “la EPOC es una enfermedad que suele presentarse en pacientes que tienen otras comorbilidades. Edad y comorbilidades son dos factores que por sí solos predisponen a la covid-19 y a padecerla de forma más grave. Además, ahora tenemos evidencia de que la EPOC también puede conducir a una evolución clínica de mayor gravedad. Por eso, debemos poner el foco en que estos pacientes sigan las recomendaciones preventivas frente a la pandemia”.
Vivir con nuevos aires
Dentro de las numerosas iniciativas puestas en marcha con motivo del Día Mundial de la EPOC, destaca la campaña #ParaVivirConAire, que, a través de las experiencias de pacientes, quiere trasladar a la población cómo viven su enfermedad. Es fruto de la colaboración entre la Federación Española de Pacientes Alérgicos y con Enfermedades Respiratorias (Fenaer) y la compañía AstraZeneca y su objetivo es aumentar la concienciación de una enfermedad tan desconocida que casi ocho de cada diez pacientes no están diagnosticados. Se estima que el 10% de la población española mayor de 40 años tendría EPOC, y que entre un 75% y un 80% de los pacientes no estarían diagnosticados.
La campaña se apoya en historias gráficas y vídeos con los testimonios de pacientes y de profesionales sanitarios, que explican sus esfuerzos diarios por llevar una vida mejor. En ella el psiquiatra Luis Gutiérrez Rojas hace hincapié en las repercusiones psicológicas de la EPOC, una faceta muy desconocida pues se estima que más de uno de cada cuatro experimenta síntomas de ansiedad y depresión.
Mariano Pastor Sanz, presidente de Fenaer, incide en la necesidad de una mayor visibilidad: “El paciente con EPOC a menudo se siente muy solo y lo que necesita es un abordaje integral que incluya no solo los tratamientos farmacológicos, sino también la rehabilitación pulmonar y el apoyo psicológico”.
En esta línea, Ana Pérez, directora médica y de Asuntos Regulatorios de AstraZeneca, explica que iniciativas como #ParaVivirConAire son el mejor ejemplo de “nuestro compromiso con la sociedad y, más específicamente, con los pacientes que sufren EPOC. En AstraZeneca, contamos también con la campaña EnEPOCElCeroCuenta, con información útil sobre la enfermedad y consejos prácticos para llevar una vida activa y huir del sedentarismo”.
Para lograr ese abordaje integral, es fundamental cuidar no solo de su salud física, sino también de la mental. Gutiérrez Rojas, psiquiatra de la Unidad de Hospitalización en Salud Mental del Hospital Clínico San Cecilio, de Granada, destaca que cualquier tipo de enfermedad crónica como la EPOC genera “un cierto sentido de frustración que, en el caso de esta patología, se suma a la culpabilidad por su relación con el tabaco. Los estudios apuntan que uno de cada cuatro pacientes con EPOC va a sufrir ansiedad o depresión”. Por eso, “es fundamental trabajar para dotar al paciente de herramientas que le permitan convivir con la enfermedad de la mejor manera posible y evitar sus efectos psicológicos. Cuando un paciente está bien asesorado puede afrontar la enfermedad de forma más sana, incluso en tiempos de pandemia”.
José Luis Izquierdo Alonso, profesor titular de Medicina de la Universidad de Alcalá y jefe de Neumología del Hospital de Guadalajara, añade que “una buena salud mental va a permitir que colabore mejor en los programas de rehabilitación y actividad física y aumente la adhesión a los tratamientos”.
En el contexto actual de la pandemia, en el que, según el Centro de Coordinación de Alertas y Emergencias Sanitarias (CCAES), el 22% de los pacientes fallecidos por la covid-19 en España sufrían EPOC, el presidente de Fenaer indica que una encuesta realizada por su federación reveló que, durante la primera ola de la pandemia, “casi el 50% de los pacientes que habían visto sus consultas canceladas seguían aún pendientes de una nueva fecha. Hay pacientes que pueden estar un año y medio sin visitar al especialista, con los consiguientes riesgos sobre el control de su enfermedad”.
Coughs Can Send Coronavirus Virus Farther Than 6 Feet
Droplets from a cough may travel farther than 6 feet and could potentially carry enough COVID-19 virus to infect another person, according to a new study.
Public health officials have urged everyone to practice social distancing during the COVID-19 pandemic, which means leaving some space between yourself and people you don’t live with. This new study adds to other research that finds that these guidelines ― the CDC advises leaving at least 6 feet between yourself and other people ― may not go far enough.
In the latest study, researchers in Singapore estimated how droplets of various sizes might travel from a person coughing to a person standing either 1 meter or 2 meters away, or 3.2 feet and 6.5 feet away, respectively.
The study found that at a distance of about 3 feet, a person standing in front of a simulated cough really gets a direct hit of virus, becoming covered with about 65% of all the droplets generated by that cough. Most of the droplets that reach the bystander are the fat, wet kind that are heavy and fall to the floor and nearby surfaces after a sneeze or cough. The study authors write that those droplets carry a “tremendous” viral load.
When the cougher and the bystander are 2 meters, or 6.5 feet, apart, fewer of the larger droplets reach the other person, but a cough still delivers just enough virus to potentially infect the other person.
But in both cases, the droplets traveled down from the nose and mouth of the person coughing, so most fell on the lower body of the bystander. Because of this, the researchers think people probably don’t inhale these larger droplets directly, but may pick them up on their skin or clothing and then infect themselves when they pick that virus up on their hands and touch their face.
“Obviously, you don’t get sick from virus landing on your clothing. You would have to breathe it in or you would have to, you know, rub your hands all over your pants and pick up enough virus then touch your nose, eyes, or stick your finger in your mouth,” says Linsey Marr, PhD, a civil and environmental engineer at Virginia Tech, who has been studying aerosol transmission of the virus. “I don’t think that’s a big risk for transmission with this virus.”
Certainly, she says, sneezes and coughs can be dangerous, but two people would have to be really close together, almost face-to-face, to pass the virus this way.
“It has to be right in your face. You have to be really close, and those droplets fly like mini cannonballs and would have to land in your eyes, in your nostrils, which point down, so it seems less likely, or on your lips,” Marr says.
Viruses can become airborne two ways: In big, heavier droplets that are expelled from the nose or mouth, and through aerosols, droplets that are so small that they dry out quickly in air. Larger droplets contain more copies of the virus, but they are also heavy and fall to the floor and nearby surfaces quickly after a sneeze or cough. Smaller aerosols, which can be generated from the back of the throat when we talk, speak, or even sing, dry out before they reach the floor, and that allows them to continue to float in the air for minutes or even hours, and may still be present after a person leaves a room.
The researchers say that shorter people, like young children and teens, may be more at risk when a taller adult coughs, since their faces are more in line with the stream of these droplets. They say that during the pandemic, shorter people may want to stay more than 6 feet away from taller people for that reason.
The new findings support earlier work from researchers at the Massachusetts Institute of Technology, which suggested that particles from a cough, buoyed by the warm air in our breath, could travel much farther than 6 feet in a turbulent gas cloud.
And because the smaller aerosols ― which are generated just by talking or singing ― can travel over longer distances, everyone should leave as much space between themselves and others as feasible.
Guidelines like the CDC’s are best guesses, and researchers have been trying to figure out exactly how the virus moves from person to person through the air.
“It’s a good guideline, but we shouldn’t think of 6 feet as a magic number. The farther the better,” Marr says.
As researchers continue to unravel the mysteries of how people catch COVID-19, public health experts say it’s important to take a layered approach to safety. No one strategy can fully protect you on its own. That’s why it’s important to take all recommended precautions to keep yourself safe from the virus, including washing and sanitizing your hands, wearing a face mask, limiting travel and social interactions, and keeping space between yourself and others.
WebMD Health News © 2020
Cite this: Coughs Can Send Coronavirus Virus Farther Than 6 Feet – Medscape – Nov 05, 2020.
De mãos dadas, Covid-19 e a tuberculose

Um dos tópicos importantes envolvendo a recentemente descrita síndrome respiratória Covid-19, associada ao coronavírus SARS-CoV-2, envolve a coinfecção com outras doenças respiratórias, especialmente a tuberculose (TB). A estimativa é de que aproximadamente 4 milhões de indivíduos morrem anualmente por infecções do trato respiratório inferior, e considera-se que cerca de 1,4 milhões morrem unicamente devido a TB, sendo esta considerada até março de 2020 a doença infecciosa com maior número de mortes diárias em todo mundo, sendo, então, sobreposta pela Covid-19. Mas então, atualmente, qual o cenário quando temos a pandemia de Covid-19 sobrepondo os amplos e significativos números já conhecidos da TB?
De maneira semelhante ao ocorrido durante o surto anterior de Ébola (2014-2015), no qual observamos que a interrupção da continuidade de serviços essenciais, incluindo os programas nacionais para controle do HIV, TB e malária, resultou em maior número de mortes por essas patologias, que sobrepos o número de mortes pelo próprio Ébola. Observou-se a interrupção do acesso de pacientes ao tratamento para TB e HIV pois as instituições de saúde, equipes médicas e laboratórios direcionaram suas energias e recursos para o controle do surto de Ébola. O mesmo tem sido observado com a pandemia da Covid-19, mas em escala ainda maior, global.
Consequências da pandemia para tuberculose
Até o final de 2019, apenas três patógenos figuravam como prioridade na lista da Organização Mundial da Saúde (OMS) para pesquisa e desenvolvimento: Mycobacterium tuberculosis e os dois coronavírus previamente descritos — SARS-CoV (2002) e MERS-CoV (2013). Com a emergência da atual pandemia, SARS-CoV-2 foi obrigatoriamente adicionada a essa listagem.
A análise de um modelo de estudo divulgado pelo programa Stop TB Parternship da OMS, considerando os 20 países prioritários que representam 54% dos casos mundiais de TB, indicam que a pandemia pela Covid-19 terá consequências drásticas para os serviços de controle de TB.
As medidas de lockdown, medo da infecção pelo novo coronavírus por parte dos pacientes e prioridades nas políticas públicas e das instituições de saúde para o combate ao SARS-CoV-2 têm resultado em limitações significativas sobre o diagnóstico, tratamento e serviços de prevenção da TB, levando a uma expectativa de comprometimento de todo o progresso obtido no combate a TB nos últimos 5 anos.
Dessa forma, espera-se também um aumento importante no número de casos e mortes por TB nos próximos 5 anos. As estimativas indicam que sob a política de lockdown por 3 meses e 10 meses para e reestruturação dos serviços de saúde, o mundo pode assistir a 6,3 milhões de casos adicionais de TB entre 2020 e 2025, e um adicional de 1,4 milhões de óbitos por TB no mesmo período. Não é coincidência de que as regiões geográficas com as piores e devastadoras consequências da Covid-19 sejam aquelas também com alta incidência de TB. Um verdadeiro desastre. Para piorar esse cenário, o risco de erros diagnósticos em favorecimento da Covid-19, a subvalorização da coinfecção, e o tratamento com imunossupressão da resposta celular com possível exacerbação da tuberculose agravam os problemas da situação atual que enfrentamos.
Coinfecção
Assim como é provável que as medidas de Saúde Pública implementadas com o isolamento/distanciamento social exacerbem o estigma e as barreiras aos direitos humanos por comunidades marginalizadas, com consequências deletérias importante à Saúde Mental em populações específicas. Adicionalmente, espera-se que os pacientes com TB ativa, coinfecção TB-HIV ou doença pulmonar crônica, e venham a adquirir infecção por Covid-19 desenvolverão um quadro clínico que requer especial atenção, e vice-versa, inclusive quanto as sequelas que vão exigir medidas de reabilitação individualizadas e prolongadas, aumentando os custos gerais com essas doenças.
A tuberculose não desapareceu, e desconhece-se as complicações e mortalidade na coinfecção. Estudos anteriores na coinfecção da TB com influenza indicaram maior risco de morbidade, hospitalização e mortalidade na associação dessas doenças quando comparadas com a monoinfecção. O mesmo pode-se esperar da coinfecção Covid-19 e tuberculose.
Tais observações ressaltam a importância da manutenção dos programas e serviços de controle da TB de forma a evitar outras calamidades. Mesmo frente as necessidades de adaptações, é importante que os programas anti-TB se reformulem para estratégias como telemedicina, saúde informatizada e monitoramento efetivo de infecção por TB em comunidades para fornecer o cuidado adequado aos pacientes sintomáticos respiratórios afetados pela TB.
Consequências do novo “normal”
Por outro lado, a Covid-19 paralelamente poderá contribuir indiretamente para reduzir o número de casos de infecções respiratórias por outros patógenos, inclusive M. tuberculosis. O uso de máscaras, a quarentena com distanciamento social e a lavagem de mãos possivelmente vão alterar o padrão de transmissibilidade de doenças. Por que não aproveitar a oportunidade atual para elaborar estratégias efetivas de vigilância, rastreio, diagnóstico e manejo terapêutico que tenham efeito sobre o controle das doenças infecciosas respiratórias?
Algum exemplo de sucesso nesse direcionamento para combate a diferentes patógenos tem sido observado, como através do desenvolvimento de terapias que modulam a resposta imune do hospedeiro. Estão em andamento os ensaios clínicos em fase I e II sobre o uso de anti-interleucina (IL) 1, anti-IL-6R, e terapias celulares com células tronco-mesenquimais alogênicas. Dessa forma, espera-se que a segurança das modalidades de tratamento anti-Covid-19 para controle da inflamação e restauração do tecido pulmonar podem também ter relevância para o tratamento da fisiopatologia da tuberculose.
Outros detalhes sobre as questões diversas dessa discussão podem ser observados nas referências abaixo.
Autor(a):
Rafael Duarte
M.D., PhD. ⦁ Médico ⦁ Microbiólogo ⦁ Professor Associado / Lab. Micobactérias, Depto. Microbiologia Médica, Instituto de Microbiologia Paulo de Góes, Centro de Ciências da Saúde – Universidade Federal do Rio de Janeiro
Referências bibliográficas
- Bandyopadhyay A, Palepu S, Bandyopadhyay K, Handu S. Covid-19 and tuberculosis co-infection: a neglected paradigm. Monaldi Arch Chest Dis. 2020 Sep 4;90(3).
- Chopra KK, Arora VK, Singh S. Covid 19 and tuberculosis. Indian J Tuberc. 2020 Apr;67(2):149-151.
- Jain VK, Iyengar KP, Samy DA, Vaishya R. Tuberculosis in the era of Covid-19 in India. Diabetes Metab Syndr. 2020 Sep-Oct;14(5):1439-1443.
- Khurana AK, Aggarwal D. The (in)significance of TB and Covid-19 co-infection. Eur Respir J 2020; 56: 2002105.
- McQuaid CF, McCreesh N, Read JM, Sumner T; CMMID Covid-19 Working Group, Houben RMGJ, White RG, Harris RC. The potential impact of Covid-19-related disruption on tuberculosis burden. Eur Respir J. 2020 Aug 13;56(2):2001718.
- Saunders MJ, Evans CA. Covid-19, tuberculosis and poverty: preventing a perfect storm. Eur Respir J. 2020 Jul 9;56(1):2001348.
- Tadolini M, García-García JM, Blanc FX, Borisov S, Goletti D, Motta I, Codecasa LR, Tiberi S, Sotgiu G, Migliori GB; GTN TB/Covid group. On tuberculosis and Covid-19 co-infection. Eur Respir J. 2020 Aug 20;56(2):2002328.
- Yang H, Lu S. Covid-19 and Tuberculosis. J Transl Int Med. 2020 Jun 25;8(2):59-65. doi: 10.2478/jtim-2020-0010.
Qual aplicativo de celular pode ser mais eficaz na cessação de tabagismo?

O tabagismo é uma importante causa de morte no mundo, sendo responsável por mais de uma a cada dez. Apesar de necessário, existem pouca demanda para tratamentos presenciais de cessação do fumo, por isso, diversos aplicativos de celular têm sido utilizados em todo o mundo.
Com o objetivo de determinar a eficácia desses aplicativos, um estudo realizado nos Estados Unidos teve seus resultados publicados, no último mês, no JAMA Internal Medicine.
Aplicativos para cessação do tabagismo
O ensaio clínico duplo-cego e randomizado comparou dois aplicativos para smartphone: o iCanQuit, que utiliza a terapia de aceitação e compromisso versus o QuitGuide, aplicativo do National Cancer Institute que utiliza as diretrizes de prática clínica dos EUA.
Os participantes foram recrutados online, foram randomizados e completaram pesquisas de acompanhamento em 3, 6 e 12 meses após a randomização.
O desfecho primário foi autorrelato de abstinência de prevalência (PPA) de 30 dias 12 meses após a randomização. Os secundários foram: abstinência de prevalência de sete dias em 12 meses, abstinência prolongada, abstinência de prevalência de 30 dias e de sete dias em três e seis meses após a randomização e dados ausentes com imputação múltipla ou codificados como tabagismo e cessão de produtos de tabaco (incluindo cigarros eletrônicos) em 12 meses.
Resultados
Após exclusões, 2.415 pessoas foram incluídas no estudo: 1.214 para o grupo iCanQuit e 1.201 para o QuitGuide. As taxas de retenção de dados de acompanhamento foram 86,7% (2.093 de 2.415) em geral em 3 meses (iCanQuit, 85,9% [1.043 de 1214] vs QuitGuide, 87,4% [1.050 de 1201]; P = 0,20), 88,4% (2136 de 2415) no geral em 6 meses (iCanQuit, 87,1% [1058 de 1214] vs QuitGuide, 89,8% [1078 de 1201]; P = 0,05) e 87,2% (2.107 de 2415) no geral em 12 meses (iCanQuit, 85,7% [1040 de 1214] vs QuitGuide, 88,8% [1067 de 1201]; P = 0,02]) .
A idade média foi de 38,2 anos. Mais de 70% eram mulheres (1.700), 69% brancos (1.666) e 35% minorias étnicas e raciais (868). Entre os participantes, 83% (2.009) fumavam por dez anos ou mais e 74% (1.803) fumavam mais de meio maço por dia (≥11 cigarros).
Sobre o desfecho primário, os participantes do iCanQuit tiveram 1,49 vezes mais chances de parar de fumar em comparação com os participantes do QuitGuide (28,2% [293 de 1040] vs 21,1% [225 de 1067]; chances razão [OR], 1,49; IC 95%, 1,22-1,83; P <0,001).
Os resultados dos desfechos secundários foram semelhantes e todos foram estatisticamente significativos para:
- PPA de sete dias no acompanhamento de 12 meses (OR, 1,35; IC de 95%, 1,12-1,63; P = 0,002);
- Abstinência prolongada no seguimento de 12 meses (OR, 2,00; IC 95%, 1,45-2,76; P <0,001);
- Abstinência de todos os produtos do tabaco no acompanhamento de 12 meses (OR, 1,60; IC 95%, 1,28- 1,99; P <0,001);
- PPA de 30 dias em acompanhamento de três meses (OR, 2,20; IC de 95%, 1,68-2,89; P <0,001);
- PPA de 30 dias em acompanhamento de seis meses (OR, 2,03; IC de 95%, 1,63-2,54; P <0,001);
- PPA de sete dias no acompanhamento de três meses (OR, 2,04; IC de 95%, 1,64-2,54; P <0,001);
- PPA de um dia no acompanhamento de 6 meses (OR, 1,73; IC de 95%, 1,42-2,10; P <0,001).
Em comparação com as pessoas que usaram o QuitGuide, os participantes do iCanQuit abriram o aplicativo mais vezes (média de 37,5 [88,4] vs 9,9 [50,0]; P <0,001), tiveram mais minutos gastos por sessão (média de 3,9 [5,3 ] vs 2,6 [2,6] minutos; P <0,001) e mais dias de uso único (média de 24,3 [50,2] vs 7,1 [15,8] dias; P <0,001).
Além disso, os usuários do iCanQuit relataram maior satisfação geral (865 de 977 [88,5%] vs 773 de 1002 [77,1%]; P <0,001), acharam mais útil (805 de 1005 [80,1%] vs 739 de 1033 [71,5%]; P <0,001) e foram mais propensos a recomendar o aplicativo a um amigo (840 de 1011 [83,1%] vs 724 de 1024 [70,7%]; P <0,001).
Conclusões
Por estarem presentes no dia a dia do paciente, aplicativos podem ser uma opção útil no acompanhamento de tratamentos ou até mesmo uma forma de intervenção significativa. Entre os aplicativos que podem auxiliar tabagistas que desejam ou precisam parar de fumar, o iCanQuit, que utiliza a terapia de aceitação e compromisso, foi mais eficaz que aquele que usa diretrizes de prática clínica estadunidenses.
É importante encontrar opções brasileiras de aplicativos que utilizem a mesma metodologia e para indicá-los no tratamento dessas pessoas.
*Esse artigo foi revisado pela equipe médica da PEBMED
Referência bibliográfica:
- Bricker JB, et al. Efficacy of Smartphone Applications for Smoking Cessation: A Randomized Clinical Trial. JAMA Intern Med. Published online September 21, 2020. doi: 10.1001/jamainternmed.2020.4055 https://jamanetwork.com/journals/jamainternalmedicine/article-abstract/2770816
#Los #enfermos respiratorios sí deben llevar #mascarilla
Un análisis internacional de neumólogos aconseja, en contra de lo recomendado, que los enfermos de asma o EPOC se protejan con mascarillas.

Desde que se estableciera la obligatoriedad de llevar mascarilla en todos los espacios de uso público para frenar la pandemia del coronavirus, en las consultas de Atención Primaria han proliferado las solicitudes de certificados para justificar la exención del uso de esa prenda por razones de salud.
La normativa sobre este aspecto contempla una serie de excepciones a la obligación de usar mascarilla, como el hecho de que las personas presenten algún tipo de enfermedad o dificultad respiratoria. Una Orden del 20 de mayo de 2020 establece que “se exceptúa de esta obligación a aquellas personas que presenten algún tipo de dificultad respiratoria que pueda verse agravada por la utilización de la mascarilla y a aquellas cuyo uso se encuentre contraindicado por motivos de salud o discapacidad.”
En el Respiratory Effectiveness Group, un grupo de médicos e investigadores de España, Reino Unido, Estados Unidos, Canadá y Australia, entre otros países, han revisado la evidencia disponible. Sus exhaustivas investigaciones han dado lugar a un estudio que se publica hoy en European Respiratory Journal. El análisis concluye que no utilizar mascarilla para evitar un ataque de asma o exacerbaciones de la EPOC o de otras enfermedades respiratorias debido a un supuesto aumento de las presiones inspiratorias a través de una mascarilla está infundado. No hay evidencia que sustente esta excepción y sí, en cambio, que los pacientes con una enfermedad respiratoria tienen un alto riesgo de sufrir covid-19 grave.
Eliminar la excepción
De ahí que los autores insten a los gobiernos a eliminar esta excepción. “Los pacientes con asma, EPOC u otras enfermedades respiratorias deben usar la mascarilla sin excepciones. Si tienen insuficiencia respiratoria o una sensación de falta de aire al ponérsela, lo más recomendable es que limiten su actividad, especialmente al aire libre”, indica Joan B. Soriano, epidemiólogo del Servicio de Neumología del Hospital La Princesa en Madrid, miembro del Covid-19 Clinical Network Knowledge Exchange Team de la OMS en Ginebra, y autor principal del estudio.
Sinthia Bosnic-Anticevich, del Instituto Woolcock de la Universidad australiana de Sidney, presidenta del REG y también coautora, añade: “Si usted o un familiar suyo padecen asma, EPOC u otra enfermedad respiratoria crónica, use mascarilla para protegerse y proteger a los demás. Este es un virus nuevo y una enfermedad nueva, pero la información es clara y las decisiones individuales son fundamentales para derrotar esta pandemia”.
Se estima que hay 545 millones de personas en todo el mundo que padecen una enfermedad respiratoria crónica, y no usar mascarilla puede conllevar un mayor riesgo de infección personal y grupal. “A falta de una vacuna eficaz debemos extremar al máximo las medidas de prevención, incluyendo el uso apropiado de las mascarillas. Esto incluye también y en especial a los pacientes respiratorios”, comenta Marc Miravitlles, neumólogo del Hospital Universitario Vall d’Hebron, en Barcelona.