Últimas notícias do evento
Strep A and Tic Worsening: Final Word?

Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one third of whom also had attention deficit hyperactivity disorder (ADHD) and one third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online February 10 in Neurology.https://6ca5e71ac1dd37b9cb45471ee9ba2603.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
“Intense Debate”
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) — a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary, Calgary, Canada, in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit
- Weekly diaries; parents were asked to indicate in the diary worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report by phone or email any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of ≥ 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into 4 categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposurehttps://6ca5e71ac1dd37b9cb45471ee9ba2603.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
Unlikely Trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs, and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or Cornerstone?
Commenting on the study for Medscape Medical News, Margo Thienemann, MD, clinical professor of psychiatry, Stanford University School of Health, Stanford, California, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham Medical School, UK, and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Missouri, suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick — a cornerstone?— in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary, the Michael P Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Cavanna, Coffman, and Thienemann have disclosed no relevant financial relationships.
Neurology. Published online February 10, 2021. Abstract, Editorial
Medscape Medical News © 2021
Cite this: Strep A and Tic Worsening: Final Word? – Medscape – Feb 12, 2021.
Novo caminho para a estimativa de risco de #câncer de mama na população geral

Novos achados de mutações genéticas no câncer de mama em mulheres sem história familiar da doença abrem um novo caminho para a estimativa de risco e podem modificar a forma como essa população é orientada.
Os achados são provenientes de dois grandes estudos, ambos publicados em 20 de janeiro no periódico The New England Journal of Medicine.
Os dois artigos são “extraordinários” por ampliarem e validarem o painel genômico como forma de contribuir para o rastreamento de mulheres com risco de câncer de mama, comentou o Dr. Eric Topol, médico, professor de medicina molecular, Scripps Research, nos Estados Unidos, e editor chefe do Medscape.
“Tradicionalmente, os exames genéticos para identificar genes hereditários do câncer de mama são focados em mulheres de alto risco que apresentam importante história familiar de câncer de mama ou nas que foram diagnosticadas ainda jovens, por exemplo, antes dos 45 anos”, comentou o primeiro pesquisador de um dos estudos, Dr. Fergus Couch, Ph.D., patologista na Mayo Clinic, nos EUA.
“Embora o risco de câncer de mama geralmente seja menor para mulheres sem história familiar da doença (…) ao avaliarmos todas as mulheres nós descobrimos que 30% das mutações do câncer de mama ocorreram nas que não apresentavam alto risco”, explicou o pesquisador.
Em ambos os estudos, oito genes (BRCA1, BRCA2, PALB2, BARD1, RAD51C, RAD51D, ATM e CHEK2)apresentaram mutações ou variantes significativamente associadas a risco de câncer de mama.
No entanto, a distribuição das mutações entre mulheres com câncer de mama diferiu da distribuição entre mulheres sem diagnóstico de câncer, observou o Dr. Steven Narod, médico do Women’s College Research Institute, no Canadá, em um editorial que acompanhou a publicação.
“Para os médicos, isso significa que agora que estamos expandindo o uso do painel genético para incluir mulheres sem diagnóstico de câncer de mama com risco moderado de desenvolver a doença no que tange a história familiar, passaremos mais tempo orientando mulheres com mutações CHEK2 e ATM“, escreveu. Hoje em dia, esses dois genes “são agrupados aos demais (…) A maior parte da discussão pré-teste atualmente está focada nas implicações de encontrar uma mutação BRCA1 ou BRCA2.“
Segundo o Dr. Steven, os novos achados podem levar a novas estratégias de manejo de risco. “A maioria dos tumores de mama em mulheres com mutação no ATM ou CHEK2 são positivos para receptores de estrogênio, então essas mulheres podem ser candidatas a terapias antiestrogênicas como tamoxifeno, raloxifeno ou inibidores de aromatase”, escreveu.
O Dr. Steven observou que, por ora, o acompanhamento da maioria das mulheres com qualquer uma das mutações vai consistir apenas em rastreamento, começando com ressonância magnética (RM) aos 40 anos.
A comunidade médica ainda não está pronta para expandir o rastreamento genético para a população em geral, alertou o Dr. Walton Taylor, médico e ex-presidente da American Society of Breast Surgeons (ASBrS).
A ASBrS atualmente recomenda que todas as pacientes com câncer de mama, assim como aquelas em alto risco de desenvolver a doença, realizem os exames genéticos. “Todas as mulheres em risco devem ser testadas, e todas as pacientes com variantes patogênicas precisam ser tratadas adequadamente – isso salva vidas”, enfatizou o Dr. Walton.
No entanto, “pessoas sem diagnóstico de câncer e sem história familiar não precisam de exames genéticos nesse momento”, ele disse ao Medscape.
Quanto ao que os médicos podem fazer para cuidar melhor de pacientes com mutações que predispõem ao câncer de mama, o Dr. Walton disse que “é surpreendentemente simples”.
Todos os laboratórios genéticos têm pessoas que realizam o aconselhamento genético para orientar os próximos passos das pacientes, apontou o Dr. Walton, e [nos EUA,] a maioria dos pacientes com câncer é assistida por enfermeiras treinadas para esse tipo de orientação, que se certificam que esses pacientes sejam devidamente testados e acompanhados.
Os membros da ASBrS seguem as diretrizes da National Comprehensive Cancer Network quando identificam portadoras de uma variante patogênica. O Dr. Walton disse que essas diretrizes são muito úteis para virtualmente todas as mutações identificadas até agora.
“Essa pesquisa não é necessariamente nova, mas atesta o que estamos fazendo, e isso contribui para sabermos que estamos seguindo o caminho certo”, disse o Dr. Walton. O trabalho “confirma que o que achamos que está certo, está realmente certo — e isso importa”, afirmou.
Achados do consórcio CARRIERS
O estudo, liderado pela equipe da Mayo Clinic e conduzido pelo consórcio Cancer Risk Estimates Related to Susceptibility (CARRIERS), realizou a análise de dados de 17 estudos epidemiológicos avaliando mulheres na população em geral que tiveram câncer de mama. Para o estudo, que foram realizados nos Estados Unidos, foram sequenciadas variantes patogênicas de 28 genes que predispõem ao câncer em 32.247 pacientes com câncer de mama (casos) e em 32.544 mulheres sem diagnóstico da doença (controles).
Na análise global do CARRIERS, a prevalência de variantes patogênicas em 12 genes clinicamente acionáveis foi de 5,03% entre os casos e de 1,63% entre os controles. A prevalência foi semelhante em mulheres brancas não hispânicas e negras não hispânicas e hispânicas, tanto entre os casos quanto entre os controles. A prevalência entre pacientes de descendência asiática foi menor, de apenas 1,64%, observaram.
Dentre pacientes que tiveram câncer de mama, as variantes patogênicas mais comuns incluíram BRCA2 (1,29% dos casos), seguida pela CHEK2 (1,08%) e BRCA1 (0,85%).
As mutações no BRCA1 aumentaram o risco de câncer de mama em mais de 7,5 vezes, e as mutações no BRCA2 aumentaram esse risco em mais de cinco vezes, observam os pesquisadores.
As mutações no PALB2 aumentaram o risco de câncer de mama em aproximadamente quatro vezes, acrescentam.
As taxas de prevalência de BRCA1 e BRCA2 para entre pacientes com câncer de mama declinaram rapidamente após a idade de 40 anos. O declínio de outras variantes, incluindo ATM, CHEK2 e PALB2, foi limitado com o aumento da idade.
Assim, mutações nesses cinco genes foram associadas a um risco absoluto de câncer de mama ao longo da vida > 20% até os 85 anos de idade entre brancas não hispânicas.
As variantes patogênicas no BRCA1 ou BRCA2 levaram a um risco ao longo da vida de câncer de mama de aproximadamente 50%. As mutações no PALB2 levaram a um risco da doença ao longo da vida de cerca de 32%.
O risco de mutação em genes específicos variou de acordo com o tipo de câncer de mama. Por exemplo, mutações nos genes BARD1, RAD51C e RAD51d aumentaram o risco de câncer de mama negativo para o receptor de estrogênio, assim como de câncer de mama triplo negativo, observaram os autores, enquanto as mutações nos genes ATM, CDH1 e CHEK2 aumentaram o risco de câncer de mama positivo para o receptor de estrogênio.
“Essas estimativas refinadas das prevalências das variantes patogênicas entre mulheres com câncer de mama na população em geral, em oposição a pacientes de alto risco específicas, podem informar discussões futuras a respeito de exames em pacientes com câncer de mama”, observaram os autores do Breast Cancer Association Consortium (BCAC).
“Os riscos de câncer de mama associados a variantes patogênicas nos genes avaliados na análise de base populacional CARRIERS também fornecem informações importantes para a avaliação do risco e o aconselhamento de mulheres com câncer de mama que não têm critérios de seleção para alto risco”, sugeriram.
Achados similares no segundo estudo
O segundo estudo foi conduzido pelo BCAC sob a liderança da Dra. Leila Dorling, Ph.D., University of Cambridge, no Reino Unido. Esse grupo sequenciou 34 genes de susceptibilidade de 60.466 mulheres com câncer de mama e 53.461 controles sem diagnóstico de câncer.
“Variantes com proteínas truncadas em cinco genes (ATM, BRCA1, BRCA2, CHEK2 e PALB2) foram associadas com um risco significativo de câncer de mama em geral (P < 0,0001)”, relataram os membros do BCAC. “Para esses genes, a razão de chances variou de 2,10 a 10,57”, acrescentaram.
A associação entre o risco geral de câncer de mama e as mutações em sete outros genes foi mais discreta, conferindo aproximadamente o dobro do risco de câncer de mama em geral, embora esse risco seja três vezes maior para mutações do TP53.
Para os 12 genes que o consórcio destacou como estando associados ou a um risco significativo ou mais discreto de câncer de mama, o tamanho do efeito não variou significativamente entre mulheres europeias e asiáticas, observaram os autores. Novamente, o risco de câncer de mama positivo para o receptor de estrogênio foi mais de duas vezes maior para aquelas que tinham mutações no ATM ou CHEK2. Ter mutações no BARD1, BRCA1, BRCA1, PALB2, RAD51C, e RAD51D conferiu maior risco de doença negativa para o receptor de estrogênio do que de doença positiva.
Também houve uma associação entre raras variantes com perda de sentido em seis genes (CHEK2, ATM, TP53, BRCA1, CDH1 e RECQL) e risco de câncer de mama em geral, com evidências mais claras para o CHEK2.
“O risco absoluto estimado coloca as variantes com proteína truncada dos genes BRCA1, BRCA2 e PALB2 na categoria de alto risco, e as variantes com proteína truncada dos genes ATM, BARD1, CHEK2, RAD51CC e RAD51D na categoria de risco moderado”, afirmaram a Dra. Leila e colaboradores.
“Esses resultados podem guiar o rastreamento, assim como a prevenção, com redução do risco de cirurgia ou de uso de medicamentos, de acordo com diretrizes nacionais”, sugeriram os autores.
O estudo CARRIERS foi financiado pelos National Institutes of Health. O estudo da Dra. Leila e colaboradores foi financiado por programas de pesquisa e inovação da European Union Horizon 2020, entre outros. O Dr. Steven informou não ter conflitos de interesses.
New Eng J Med. Publicado on-line em 20 de janeiro de 2021. Couch et al, Abstract ; Estudo do BCAC, Texto completo; Editorial
Medscape Notícias Médicas © 2021 WebMD, LLC
Citar este artigo: Novo caminho para a estimativa de risco de câncer de mama na população geral – Medscape – 11 de fevereiro de 2021.
As manifestações orais podem nortear o diagnóstico de Covid-19? — Parte 1
A Doença Coronavírus 2019 (Covid-19) se espalhou exponencialmente por todo o mundo desde sua descoberta na China no final de 2019. As manifestações típicas de Covid-19 incluem febre, tosse seca, cefaleia e fadiga. Contudo, apresentações atípicas são cada vez mais relatadas. Estudos reconheceram as lesões orais como manifestações associadas ao Covid-19, sendo que as mais comuns são as ulcerativas, vesicobolhosas e maculares. A ocorrência de manifestações orais no Covid-19 parece ser subnotificada, principalmente devido à falta de exame bucal de pacientes com suspeita e/ou confirmação diagnóstica. O exame oral de todos os casos suspeitos e confirmados é fundamental para melhor compreensão e documentação das manifestações da cavidade oral relacionadas a Covid-19.

Patogenia
Pesquisas atuais sugerem que os danos do novo coronavírus às vias respiratórias e a outros órgãos podem estar relacionados à distribuição de receptores da enzima conversora de angiotensina 2 (ECA-2). Dessa forma, células com receptor ECA-2 podem se tornar hospedeiras do vírus e causar reações inflamatórias em órgãos e tecidos relacionados, como a mucosa da cavidade oral, língua e glândulas salivares. Existem dois mecanismos que podem explicar o desenvolvimento de tais lesões: diretamente, através dos efeitos do vírus replicante, onde essas lesões serão específicas para SARS-CoV-2 e indiretamente, por meio do estresse físico e psicológico associados ao vírus ou secundário aos medicamentos utilizados em seu tratamento.
Manifestações orais relacionadas ao Covid-19
Com o crescente número de casos de Covid-19, vários relatos sobre lesões de cavidade oral têm sido publicados. Assim como nas lesões dermatológicas, a maioria dos trabalhos é de cartas ao editor ou de casos clínicos, portando de baixa qualidade científica. Não há estudos que tenham apresentado lesões patognomônicas, mas a descrição das lesões de cavidade oral já descritas na literatura poderão auxiliar ou nortear os diagnósticos.
Tipos de lesões orais já descritos em pacientes com Covid-19:
Lesões ulcerativas
As lesões ulcerativas são as manifestações orais mais frequentes relacionadas ao Covid-19. A lesões variam entre ulceras únicas, múltiplas, dolorosas ou erosões graves. O local das úlceras varia, mas o dorso da língua é o local afetado com maior frequência, seguido pelo palato duro e a mucosa bucal. Diferentes fatores, incluindo erupção por medicamentos, vasculite ou vasculopatia trombótica secundária a Covid-19, foram sugeridos como outras causas para o desenvolvimento destas lesões (Fig. 1 e 2).


Lesões vesicobolhosas e maculares
As apresentações variam entre bolhas, lesões eritematosas, lesões petequiais e eritema multiformes. Destes, as lesões do tipo eritema multiformes são a apresentação mais comum. A maioria dos casos com manifestações vesicobolhosas e maculares estão associadas a lesões cutâneas (Fig. 3 e 4).

Lesões aftosas
As lesões aftosas se manifestam como múltiplas úlceras superficiais com halos eritematosos e pseudomembranos amarelos e brancos nas mucosas queratinizadas e não queratinizadas. As lesões aftosas sem necrose são observadas em pacientes mais jovens com infecção leve, enquanto lesões aftosas com necrose e crostas hemorrágicas são observadas mais frequentemente em pacientes mais velhos, com imunossupressão e infecção grave. A regressão das lesões orais é associada à melhora da doença sistêmica. O nível elevado de fator de necrose tumoral‐α em pacientes com Covid‐19 pode levar à quimiotaxia de neutrófilos para a mucosa oral e ao desenvolvimento de lesões aftosas. Estresse e imunossupressão secundários à infecção por Covid-19 podem ser outras razões possíveis para o surgimento de tais lesões.
Continua….
Autor(a):
Vera Lucia Angelo Andrade
Graduação em Medicina pela UFMG em 1989 • Residência em Clínica Médica/Patologia Clínica pelo Hospital Sarah Kubistchek • Gastroenterologista pela Federação Brasileira de Gastroenterologia • Especialista em Doenças Funcionais e Manometria pelo Hospital Israelita Albert Einstein • Mestre e Doutora em Patologia pela UFMG • Responsável pelo Setor de Motilidade da Clínica SEDIG BH desde 1995 • http://lattes.cnpq.br/0589625731703512
Em conjunto com: Elias Felipe Romanos da Matta¹.
¹ Especialista em Clínica Médica pelo Hospital Felício Rocho. Especializando em Gastroenterologia e Nutrologia no Hospital Vera Cruz.
Referências bibliográficas:
- Halboub, Esam, Al-Maweri, Sadeq Ali, Alanazi, Rawan Hejji, Qaid, Nashwan Mohammed, & Abdulrab, Saleem. (2020). Orofacial manifestations of Covid-19: a brief review of the published literature. Brazilian Oral Research, 34, e124. Epub October 30, 2020. doi: doi: 10.1590/1807-3107bor-2020.vol34.0124
- Iranmanesh, B, Khalili, M, Amiri, R, Zartab, H, Aflatoonian.(2020). M. Oral manifestations of Covid‐19 disease: A review article. Dermatologic Therapy. e14578. doi: 10.1111/dth.14578
- Anne-Gaëlle Chaux-Bodard, Sophie Deneuve and Aline Desoutter. (2020).Oral manifestation of Covid-19 as an inaugural symptom? J Oral Med Oral Surg, 26 2 18. doi: 10.1051/mbcb/2020011
- Amorim Dos Santos, J., Normando, A., Carvalho da Silva, R. L., De Paula, R. M., Cembranel, A. C., Santos-Silva, A. R., & Guerra, E. (2020). Oral mucosal lesions in a Covid-19 patient: New signs or secondary manifestations?. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases, 97, 326–328. doi: 10.1016/j.ijid.2020.06.012
- Soares, CD, Carvalho, RA, Carvalho, KA, Carvalho, MG, & Almeida, OP (2020). Carta ao Editor: Lesões orais em um paciente com Covid-19. Medicina oral, patologia oral y cirugia bucal , 25 (4), e563 – e564. doi: 10.4317/medoral.24044
- Cruz Tapia, R. O., Peraza Labrador, A. J., Guimaraes, D. M., & Matos Valdez, L. H. (2020). Oral mucosal lesions in patients with SARS-CoV-2 infection. Report of four cases. Are they a true sign of Covid-19 disease?. Special care in dentistry : official publication of the American Association of Hospital Dentists, the Academy of Dentistry for the Handicapped, and the American Society for Geriatric Dentistry, 40(6), 555–560. doi: 10.1111/scd.12520
- Díaz Rodríguez M, Jimenez Romera A, Villarroel M. Manifestações orais associadas com Covid-19. Doenças orais. 2020 Jul. doi: 10.1111/odi.13555
Lengua, palma de la mano y planta del pie, las nuevas “víctimas” de la COVID-19

A un año del primer caso de COVID-19 en España, esta enfermedad mantiene intacta su capacidad de sorprender al colectivo médico. Cada día está más claro que los efectos de la infección provocada por el SARS-CoV-2 van mucho más allá de la pérdida de olfato y los daños pulmonares, como se muestra en un estudio liderado por el Hospital Universitario de la Paz, publicado recientemente en British Journal of Dermatology.[1]
En este estudio con 666 pacientes con COVID-19 ingresados en el hospital de campaña de Ifema entre el 10 y el 25 de abril de 2020 con neumonías leves o moderadas, y cuya primera autora es la Dra. Almudena Nuño-González, dermatóloga del Hospital Universitario de La Paz, se observó que una parte significativa de ellos sufrió lesiones mucocutáneas, orales y palmoplantares.
Este estudio, pionero a nivel mundial, realizado por profesionales del Hospital Universitario de La Paz y de Atención Primaria del Servicio Madrileño de Salud (SERMAS) de la Comunidad de Madrid, sirvió para corroborar las declaraciones previas del Dr. Tim Spector, profesor de epidemiología genética en el King’s College London, en Londres, Inglaterra, que a mediados de enero advirtió que “una de cada cinco personas con COVID-19 presenta síntomas poco comunes que no se incluyen en la lista oficial del Public Health England, como erupciones cutáneas.
Además se está registrando un número creciente de ‘lengua COVID’ y extrañas úlceras bucales”.
Sin embargo, el objetivo de este estudio no era demostrar la veracidad de las afirmaciones del Dr. Spector, ya que estas se realizaron meses después de su finalización.
“Lo que en realidad buscábamos era comprobar cuáles eran las lesiones dermatológicas más frecuentes en pacientes con COVID-19. Dado que en Ifema a todos los pacientes les habían diagnosticado COVID-19, fue un lugar en el que nos resultó fácil conseguir fotografías de las lesiones cutáneas”, explicó la Dra. Pilar Martín-Carrillo, del Consultorio Colmenarejo del SERMAS, participante también en la investigación.
Alteraciones orales en pacientes con COVID-19
El promedio de edad de los 666 pacientes con COVID-19 en los que se realizó el estudio era de 55,67 años, con ligera predominancia femenina (58%) y casi la mitad (47,1%) era de origen latinoamericano; 304 de estos pacientes (45,65%) presentaron una o más manifestaciones mucocutáneas, 78 de ellos (25,65%) padecieron alteraciones en la mucosa oral, incluyendo papilitis lingual transitoria (11,5%), glositis con marcas de los dientes en los laterales (6,6%), úlceras bucales (6,9%), glositis con depapilación en parches (3,9%) y sensación de ardor (5,3%). Todas estas manifestaciones aparecieron generalmente asociadas con disgeusia.
La Dra. Nuño señaló que algunos de estos síntomas podrían estar provocados por factores ajenos al SARS-CoV-2, “como algunos fármacos o la ventilación con oxígeno, que seca la boca y puede irritar la lengua.
Sin embargo, la lengua depapilada es 100% debida a la COVID-19, porque esto no se da por ninguna circunstancia ni ningún tratamiento”, afirmó a Univadis España.
“Este es un hallazgo que puede ayudar al diagnóstico, como la pérdida de olfato o del gusto. Son síntomas muy característicos”, añadió.
Lesiones palmoplantares en pacientes con COVID-19
Por otro lado, también se detectaron alteraciones en la piel de las palmas de las manos y las plantas de los pies en 121 pacientes (39,8%), mayoritariamente descamaciones 25,3% (77 pacientes), manchitas de color rojizo o marrón en 46 de ellos (15,1%) y sensación de ardor conocida como eritrodisestesia en 7% de los pacientes al comienzo de la infección.
Otras manifestaciones observadas fueron urticaria (6,9%), sarpullidos (2,9%) y erupciones vesiculares (1,6%). Tanto la urticaria como los sarpullidos se observaron en cualquier estadio de la enfermedad, mientras que las erupciones vesiculares típicamente aparecieron en los primeros días después de haber comenzado a manifestarse los síntomas. Ambas, la urticaria y las erupciones vesiculares, fueron observadas en los pacientes más jóvenes.
Una variedad de manifestaciones clínicas
La infección por SARS-CoV-2 se ha relacionado con múltiples síntomas: respiratorios, trombóticos, neurológicos, digestivos o cutáneos. Estos últimos se han clasificado en cinco tipos: lesiones acroisquémicas, lesiones vesiculares, erupción urticarial, exantema maculopapular o lesiones livedoides. Sin embargo, hasta la publicación de este trabajo no se habían considerado las alteraciones orales ni las lesiones palmoplantares asociadas a la COVID-19.
Los autores concluyeron que la cavidad oral “se puede alterar por la enfermedad COVID-19. El edema lingual con papilitis lingual transitoria en forma de U o la glositis con depapilación en parches son signos muy característicos, al igual que la sensación de ardor en la cavidad oral. Este ardor puede aparecer también en palmas y plantas con color rojizo o descamación y manchas. Todos pueden ser signos clave para un diagnóstico precoz de esta enfermedad”.
Todos los autores han declarado no tener ningún conflicto de interés económico pertinente.
Medscape Noticias Médicas © 2021 WebMD, LLC
Citar este artículo: Lengua, palma de la mano y planta del pie, las nuevas “víctimas” de la COVID-19 – Medscape – 11 de feb de 2021.
The Challenges of Antiviral Treatments
Physician Claudette Poole doesn’t take long to rattle off a list of antiviral medications she prescribes to her patients. “There really aren’t very many,” says Poole, a pediatric infectious disease physician at the University of Alabama at Birmingham.

And for people with Covid-19, there’s just one approved for use: remdesivir, which doesn’t seem to save lives, but speeds recovery in those who do get well. Clearly, more antivirals would be nice to have — so why don’t we have them? Inventing them, it turns out, is not so easy.
Viruses rely on human cell machinery to copy themselves, so antiviral designers face a challenge: how to stop the virus without damaging the inner workings of healthy cells too. While scientists have found several solutions to the problem, the antiviral pharmacopeia still lags behind the plethora of antibiotics available to treat bacterial infections.
But as researchers build up their knowledge of viral life cycles, antivirals may be poised to catch up. Scientists are also planning for future pandemics, in the hopes of having a better selection of antivirals to try the next time around.
Here’s where antivirals stand today, and how the list might be growing.
How Do Antivirals Work?
An antiviral drug can block any of the steps a virus uses to copy itself. To do its dirty work, a virus must attach to a host cell, sneak inside and trick that cell into copying viral genes and crafting viral proteins; after that, the newly made viruses must escape to infect new targets. At each step, viral genes or proteins need to interact with various host molecules, and each of these interactions offers an opportunity for antiviral drugs. The drugs often mimic those host molecules and act as decoys to interfere with the viral life cycle and reduce its spread.https://4124fa68669c2e6429bf5d79b5331880.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
A common approach is to interfere with the copying of viral genes into DNA or RNA to form new viral genomes. Viruses frequently have their own versions of proteins, called polymerases, for this task. The polymerases add individual building blocks called nucleotides, one by one, to the new genome as it’s being built.
For example, the drug acyclovir, used to treat herpes, goes after this genome-copying step. To the virus’s polymerase, the medicine looks like just another building block — but it’s not. Once the decoy gets into the growing strand, it prevents the addition of any more nucleotides. For the virus, it’s game over.
Another drug, oseltamivir (Tamiflu) for influenza, acts at the stage of viral exit from the infected cell. The virus uses a key protein called neuraminidase to dissolve its way out, but oseltamivir sticks to the neuraminidase and stops it from working.
Since antivirals don’t eradicate viruses directly — they just stop them from spreading cell to cell or person to person — it’s up to the body’s immune system, when possible, to mop up the invaders already present. That’s why it’s important to start antiviral treatment early, while viral numbers remain low. “The faster you can take the drug, the more you can limit the virus’s ability to spread,” says virologist Mark Heise of the University of North Carolina at Chapel Hill. Tamiflu, for example, works best when taken within 48 hours of the first symptoms, helping people recover about a day faster.
Why Are There So Few Antivirals?
The number of antivirals is paltry compared with the list of bacteria-fighting antibiotics. That’s due to several factors.
For one, antibiotics were first out of the starting gate, notes Erik De Clercq, an emeritus professor of biomedicine at KU Leuven in Belgium. The first, penicillin, was discovered in 1928 and first used in a patient in 1940. In contrast, the first antiviral, idoxuridine, was developed as an anticancer agent in 1959, was reported to block viruses in 1961, and approved in 1963 to treat herpes infections of the eye. (De Clercq, a leader in early antiviral research, described his scientific journey in the Annual Review of Pharmacology and Toxicology in 2011.)
Plus, viruses are much trickier targets than bacteria, says Monica Gandhi, an infectious disease physician at the University of California, San Francisco. Bacteria are whole living cells with all the metabolic pathways they need for survival, so they offer plenty of targets for attack. They also have unique features such as cell walls that aren’t found in human cells. That means antibiotics can interfere with cell walls, or other bacteria-specific parts and processes, to kill the pathogens without harming our own cells. And because microbes evolved antibiotics to battle each other, there is a diverse array of the compounds out in nature.https://4124fa68669c2e6429bf5d79b5331880.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
In contrast, viral pathogens live inside our own cells and depend on our proteins for most of their needs, so they offer no such easy targets. And few natural antivirals exist, so scientists need to invent them from scratch, says Kathie Seley-Radtke, a medicinal chemist at the University of Maryland, Baltimore County.
Moreover, antivirals have a limited number of possible shapes. That’s because, to block a virus’s actions, they must fit into viral proteins as decoys.
The biggest challenge, says Seley-Radtke, is to ensure that the drugs don’t hurt the human hosts as well. For example, in the case of nucleotide mimics like acyclovir, wouldn’t there be a risk that they would get into the cell’s DNA as well as the virus’s?
There are ways around that problem. In the case of acyclovir, the drug that patients swallow is an inactive form, and it is mainly activated by a viral protein. “It targets the virus very nicely, while leaving host cell DNA alone,” says Poole, who reviewed the use of acyclovir and other antivirals for newborns with herpes simplex virus in the 2018 Annual Review of Virology.
There is one unfortunate similarity between antibiotics and antivirals: In both cases, pathogens can make tiny changes to their genes and proteins that leave them unharmed by the drug. “Resistance is a huge issue in antivirals,” says Poole. Doctors used to prescribe drugs called adamantanes to people with the flu, for example — but the influenza viruses circulating among people today are unaffected by the drugs. “They are no longer effective at all,” says Poole. “There’s a desperate need to find other flu antivirals.”
The one exception to the dearth of antivirals is the flourishing pharmacopeia of meds against the human immunodeficiency virus, resulting from decades of research. Gandhi says that she can select from 30 or so medications to treat her HIV-positive patients. More are on the way — and just as well, because HIV can quickly evolve resistance to any one drug.
“HIV research set the tone,” says Heise, and it’s bringing other antivirals in its wake. “We’re seeing, again, new antivirals coming out much more quickly over the last several years.” In the last five years, for example, novel medications have turned hepatitis C from a chronic to curable condition.
“I think we’ll see more drugs for acute viral infections coming forward as well,” says Heise.
How Are Antivirals Being Used Against Covid-19?
Drugs designed against one virus often work against others, because proteins such as the polymerases used to copy virus genomes are similar across a wide range of viruses. But the current scourge requires more than the usual amount of cleverness from antiviral designers.
“Coronaviruses are pretty tricky,” Seley-Radtke says. Simple nucleotide mimics like acyclovir won’t work, because these viruses have another protein that acts as an editor, monitoring the polymerase’s work, recognizing the decoy and cutting it out.
Enter remdesivir, which had already been tested in people with Ebola (though it didn’t help them much). It’s a nucleotide mimic, but it’s a bit special. Once incorporated into a piece of the new viral genome — which in the case of coronaviruses is made of RNA — it doesn’t stop the strand’s growth right away. The polymerase keeps adding normal nucleotides. But after it’s added a few, the drug bends the RNA strand so badly that the polymerase can’t keep building. By then, though, the coronavirus editor protein no longer works; the normal nucleotides added after remdesivir seem to get in the way, says Seley-Radtke. Thus, the polymerase is stuck.
Despite that clever trick, remdesivir’s performance in people with Covid-19 has been decidedly lukewarm. On the plus side: In a trial of 1,062 people hospitalized with the virus, those who were treated with remdesivir recovered more quickly than those who received an inactive placebo. Based on this and two similar studies, the US Food and Drug Administration authorized remdesivir to treat hospitalized patients.
But what remdesivir doesn’t seem to do is save lives. In November, the World Health Organization, citing its own larger but preliminary study, recommended against remdesivir for hospitalized patients for the time being, until more research can be done. The WHO noted “no evidence that remdesivir improves survival and other outcomes.”
The results, which at first blush may seem confusing, make sense considering how antivirals work, says Poole. Because remdesivir requires multiple intravenous infusions, it’s given only to hospitalized patients. But by the time a person with Covid-19 is ill enough to be hospitalized, the virus has already run rampant across their body, so remdesivir comes too late to do much good. “The game-changer,” she says, “is going to be when we find an antiviral that you can give to people orally before they get into the hospital.”https://4124fa68669c2e6429bf5d79b5331880.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
Remdesivir’s maker, Gilead Sciences, is working on an inhaled version. And there are other antivirals in the pipeline. For example, Seley-Radtke is optimistic about another nucleotide analog, known as AT527. Under joint development by Roche and Atea, AT527 is now midway through human trials. Like remdesivir, it has delayed action, so it avoids being edited out of a growing RNA strand. But unlike remdesivir, it’s a pill you can swallow. The companies hope it can be used by hospitalized and non-hospitalized patients alike, and perhaps even be taken by people exposed to Covid-19 to prevent infection from taking hold to begin with.
The pandemic has sent scientists scrambling to find treatments. Heise, for one, is testing a wide range of drugs — not just standard antivirals — against SARS-CoV-2 in lab dishes, as part of the Rapidly Emerging Antiviral Drug Discovery Initiative (READDI). The idea is that, because the virus depends on many processes in human cells, a variety of medications that act on human proteins might give doctors an edge by hurting the virus more than the patient. That throws the doors open to considering medications that were originally designed for cancer, psychosis, inflammatory conditions and autoimmune disease, to see if they might have a shot against Covid-19.
But the READDI collaborators — including academic centers, pharmaceutical companies and nongovernmental organizations — are aiming for more than a Covid-19 treatment. READDI hopes to identify and test potential medications for as-yet-unknown infections that may crop up in the future.
By getting early human safety testing done ahead of time, they’ll be ready to spring into action when those future outbreaks happen. As Heise says, “We don’t want to repeat what we’ve just been through.”
Amen to that.
This article is part of Reset: The Science of Crisis & Recovery, an ongoing series exploring how the world is navigating the coronavirus pandemic, its consequences and the way forward. Reset is supported by a grant from the Alfred P. Sloan Foundation.
This article originally appeared in Knowable Magazine on February 9, 2021. Knowable Magazine is an independent journalistic endeavor from Annual Reviews, a nonprofit publisher dedicated to synthesizing and integrating knowledge for the progress of science and the benefit of society. Sign up for Knowable Magazine’s newsletter.123
Knowable Magazine © 2021
Cite this: The Challenges of Antiviral Treatments – Medscape – Feb 11, 2021.
Perspectives thérapeutiques dans le syndrome cardio-rénal

Antoine FAYOL*/**, Étienne PUYMIRAT***, Marine LIVROZET*, Jean-Sébastie HULOT*,*Centre d’investigation clinique **Unité médico-chirurgicale d’insuffisance cardiaque sévère ***Unité de soins intensifs de cardiologie Hôpital européen Georges Pompidou, Paris
La prise en charge du syndrome cardio-rénal va probablement évoluer dans les années à venir grâce à l’arrivée d’un nouvel arsenal thérapeutique pour les patients insuffisants cardiaques. L’objectif de ce troisième article est de présenter ces nouvelles classes thérapeutiques qui arrivent progressivement sur le marché : les gliflozines, la finérénone, le vériciguat ou l’omecamtiv mecarbil. Ils ont très probablement une place à trouver dans la prise en charge des syndromes cardio-rénaux.
Une meilleure organisation
En 2019, l’AHA recommande la création d’unités spéciales pour la prise en charge des patients ayant un syndrome cardio-rénal via la création de structures regroupant cardiologues et néphrologues pour proposer une prise en charge commune au patient(1).
L’identification d’un diagnostic étiologique, tant cardiologique que néphrologique, semble indispensable pour la prise en charge des patients et pour l’optimisation des thérapeutiques. Le bilan d’insuffisance cardiaque et d’insuffisance rénale doit donc être fait en même temps.
La création d’une consultation spécialisée avec une surveillance régulière de la volémie du patient pourrait limiter les récidives de décompensations cardiaques.
Les caractéristiques cliniques des patients dans les essais thérapeutiques portant sur le syndrome cardio-rénal nous permettent d’envisager l’intérêt de l’évaluation de nouvelles classes thérapeutiques. Pour rappel, la majorité des patients présentant un syndrome cardio-rénal sont des hommes avec des comorbidités (hypertension, diabète dans 50 à 60 % des cas, fibrillation atriale) et une cardiopathie ischémique avec une altération de la FEVG dans 50 à 60 % des cas(2,3).
Les gliflozines
Le bénéfice cardiovasculaire des gliflozines a d’abord été confirmé chez les patients diabétiques. Le tableau 1 récapitule les principaux essais thérapeutiques.
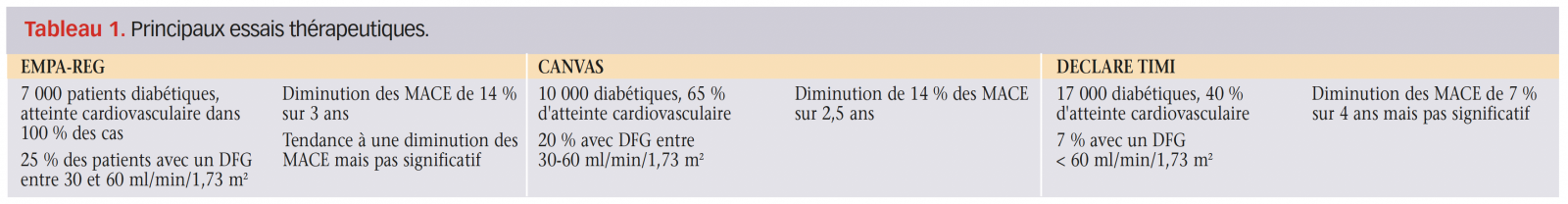
Une métaanalyse en 2019 (EMPA-REG, CANVAS, DECLARE TIMI(4-6)) s’est intéressée aux patients ayant un DFG entre 30 et 60 ml/min/1,73 m2. Pour ce groupe de patients, on observe une diminution du risque d’hospitalisation pour insuffisance cardiaque (HR 0,6), du nombre de MACE (HR 0,82) et des événements rénaux (HR 0,67).
Leurs bénéfices concernant l’insuffisance cardiaque sont également confirmés pour les patients ayant une HFrEF qu’ils soient diabétiques ou non(6,7). Dans une métaanalyse récente portant toujours sur les patients ayant une HFrEF, les patients ayant un DFG < 60 ml/min/1,73 m2 avaient une diminution significative des hospitalisations pour décompensation cardiaque et de la mortalité cardiovasculaire (HR 0,77).
Dans l’étude CREDENCE(8) (figure 1), les patients inclus étaient cette fois-ci diabétiques avec une albuminurie > 300 mg/g et un DFG entre 30 et 90 ml/ min (sous traitement conventionnel maximal). Le critère de jugement principal n’était plus cardiaque mais rénal : il s’agissait d’un critère composite d’apparition d’insuffisance rénale terminale (dialyse ou transplantation), d’un doublement de la créatinine basale ou de la mortalité cardiovasculaire et/ou rénale. L’étude a été interrompue précocement du fait d’une diminution des événements rénaux de 30 % (doublement de la créatinine ou mortalité rénale et cardiovasculaire).

Figure 1. Résultats de l’étude CREDENCE(8).
Dans l’étude DAPA-CKD (figure 2), les patients étaient diabétiques ou non avec un DFG entre 25 et 75 ml/min/1,73 m2 et une albuminurie comprise entre 0,2 g/g et 5 g/g. Le critère composite principalement rénal a lui aussi été rapidement validé (réduction de risque relatif de 37 %) avec de nouveau un arrêt précoce de l’étude(9).

Figure 2. Résultats de l’étude DAPA-CKD(9).
Enfin on peut également citer l’étude VERTIS qui s’est intéressée à l’ertugliflozine, et qui a montré qu’elle était non inférieure au placebo pour la prévention de la récidive d’événements cardiovasculaires majeurs(10).

La finérénone
La finérénone (antagoniste du récepteur aux minéralo-corticoïdes) a été évaluée chez les patients diabétiques, insuffisants rénaux dans l’étude FIDELIODKD(11) (figure 3). Au total, 5 734 patients avec un DFG compris entre 25 et 75 ml/min/1,73 m2 (avec une albuminurie > 30 mg/g) ont été inclus. Le critère de jugement principal était un critère composite associant la mortalité rénale, l’apparition d’une insuffisance rénale et une diminution de 40 % du eDFG.

Figure 3. Résultats de l’étude FIDELIO(11).
Après 2,6 ans de suivi, les patients traités par finérénone avaient une réduction significative du critère de jugement principal par rapport aux patients traités par placebo (HR 0,82 ; IC95% : 0,73-0,93 ; p = 0,001). Il y avait aussi une diminution significative du risque de survenue de MACE (AVC, infarctus du myocarde, décès d’origine cardiovasculaire) (HR 0,86 ; IC95% : 0,75- 0,99 ; p = 0,03).

Le vériciguat
Le vériciguat est un stimulateur de guanylate cyclase soluble oral, qui intervient via la voie de la guanosine monophosphate (GMP) cyclique. Il favorise la sensibilité de la guanylate cyclase soluble à l’oxyde nitrique (NO) endogène (stabilise la liaison) ce qui entraîne une vasorelaxation et une inhibition de la prolifération des muscles lisses(12).
Il a été evalué chez des patients avec une insuffisance cardiaque à FE réduite. Au total, 10 % d’entre eux avaient un DFG estimé à moins de 30 ml/min/m2 et 42 % entre 30 et 60 ml/min/m2. Le critère de jugement principal était composite associant la mortalité cardiovasculaire et la survenue d’une hospitalisation pour insuffisance cardiaque (figure 4).
Figure 4. Résultats du critère de jugement principal et dans les analyses de sous-groupe en fonction du DFG(13).
Le critère de jugement principal est survenu chez 35,5 % des patients traités et chez 38,5 % des patients ayant reçu le placebo (HR 0,90 ; IC95% : 0,82- 0,98 ; p = 0,02). Les analyses en sous-groupe n’étaient pas en faveur de l’utilisation du vériciguat chez les patients ayant un DFG < 30 ml/min/1,73 m2.

L’omecamtiv mecarbil
L’omecamtiv mecarbil(14) est un activateur direct de la myosine cardiaque, agissant directement au niveau du sarcomère, ce qui a comme effet d’augmenter le volume d’éjection systolique et de favoriser le remodelage du ventricule gauche.
Il a été évalué chez 8 256 patients avec une FEVG altérée (FEVG ≤ 35 %), ayant été hospitalisés pour un épisode de décompensation cardiaque dans l’année. Le DFG médian des patients inclus était de 58 ml/min/1,73 m2. Le critère de jugement principal était composite associant la mortalité d’origine cardiovasculaire et la survenue d’une hospitalisation pour insuffisance cardiaque.
Après 21,8 mois de suivi (figure 5), on observe une diminution significative du risque de survenue du critère composite chez les patients traités par omecamtiv mecarbil (1 523/4 120 patients [37,0 %] vs 1 607/4 112 patients [39,1 %] — HR 0,92 ; IC95% : 0,86-0,99 ; p = 0,03) ; sans diminution de la mortalité cardiovasculaire (HR 1,01 ; IC95% : 0,92- 1,11).
Figure 5. Résultats de l’étude et dans les sous-groupes d’intérêts GALACTIC-HF(14).
Les analyses en sous-groupes sont en faveur d’une efficacité plus importante du traitement chez les patients ayant une FEVG < 28 % (HR 0,84 ; IC95% : 0,77-0,92).

En pratique
▸ Une coopération entre cardiologues et néphrologues semble essentielle pour une prise en charge optimale des patients ayant un syndrome cardio-rénal. La création d’unités spéciales dédiées aux syndromes cardio-rénaux permettrait de faciliter la mise en place de thérapeutiques personnalisées.
▸ Les essais randomisés évaluant les nouvelles thérapeutiques disponibles sont nécessaires pour les patients ayant une insuffisance rénale chronique stade 4 et 5 et restent à prévoir.
Références
Cliquez sur les références et accédez aux Abstracts sur 
1. Janani R et al. Cardiorenal syndrome: classification, pathophysiology, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation 2019 ; 139 : e840-e878. Rechercher l’abstract
2. Bart BA et al. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med 2012 ; 367 : 2296-304. Rechercher l’abstract
3. Costanzo MR et al. Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure. J Am Coll Cardiol 2007 ; 49 : 675-83. Rechercher l’abstract
4. Zinman B et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015 ; 373 : 2117-28. Rechercher l’abstract
5. Lytvyn Y et al. Sodium glucose cotransporter-2 inhibition in heart failure. Circulation 2017 ; 136 : 1643-58. Rechercher l’abstract
6. Wiviott SD et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2019 ; 380 : 347-57. Rechercher l’abstract
7. McMurray JJV et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019 ; 381 : 1995-2008. Rechercher l’abstract
8. Perkovic V et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019 ; 380 : 2295-306. Rechercher l’abstract
9. Heerspink HJL et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020 ; 383 : 1436-46. Rechercher l’abstract
10. Cannon CP et al. Cardiovascular outcomes with ertugliflozin in type 2 diabetes. N Engl J Med 2020 ; 383 : 1425-35. Rechercher l’abstract
11. Bakris GL et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med 2020 ; 383 : 2219-29. Rechercher l’abstract
12. Stasch JP et al. Soluble guanylate cyclase as an emerging therapeutic target in cardiopulmonary disease. Circulation 2011 ; 123 : 2263-73. Rechercher l’abstract
13. Armstrong PW et al. Vericiguat in patients with heart failure and reduced ejection fraction. N Engl J Med 2020 ; 382 : 1883-93. Rechercher l’abstract
14. Teerlink JR et al. Cardiac myosin activation with omecamtiv mecarbil in systolic heart failure. N Engl J Med 2020 Nov 13. Rechercher l’abstract
Dieta vegetariana ou cetogênica?

Para controlar a fome, uma dieta vegetariana e com baixo teor de gordura tem vantagens em relação a uma dieta cetogênica baseada no consumo de proteína/gordura animal e com baixo teor de carboidratos, mas a dieta cetogênica ganha quando se trata de maior controle dos níveis de glicemia e insulina pós-prandiais, segundo uma nova pesquisa.
Em um estudo cruzado e cuidadosamente controlado, conduzido pelo National Institutes of Health (NIH), as pessoas consumiram menos calorias diárias quando seguiram uma dieta vegetariana e com baixo teor de gordura, mas os níveis de insulina e glicemia foram mais altos em comparação aos que fizeram uma dieta cetogênica baseada no consumo de proteína/gordura animal.
“Existe uma ideia um tanto desatualizada de que dietas com alto teor de gordura, por terem mais calorias por grama, tendem a fazer as pessoas comerem em excesso – o chamado modelo de consumo passivo excessivo”, disse ao Medscape o pesquisador sênior do estudo, Dr. Kevin Hall, Ph.D., National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) dos Estados Unidos.
O outro modelo, mais popular hoje em dia, explicou o pesquisador, é o modelo carboidrato-insulina, que preconiza que seguir uma dieta rica em carboidratos e açúcares, que causam um pico nos níveis de insulina, aumentará a fome e fará com que a pessoa coma excessivamente.
Neste estudo, Dr. Kevin Hall e colaboradores testaram essas duas hipóteses uma contra a outra.
“A resposta curta é que obtivemos resultados exatamente opostos aos do modelo de carboidrato-insulina de obesidade. Em outras palavras, em vez de as pessoas comerem mais e ganharem peso e gordura corporal, elas acabaram comendo menos com essa dieta e perdendo gordura corporal em comparação com a dieta rica em gordura”, disse o Dr. Kevin.
“Porém, o modelo de consumo passivo excessivo também falhou, porque, apesar de os participantes terem feito uma dieta muito densa em energia e rica em gordura, eles não ganharam peso nem gordura corporal. Por isso, os dois modelos sobre a razão das pessoas comerem excessivamente e ganharem peso parecem ser inadequados de acordo com o nosso estudo”, disse ele ao Medscape. “Isso sugere que as coisas são um pouco mais complicadas.”
O estudo foi publicado on-line em 21 de janeiro no periódico Nature Medicine.
Prós e contras das duas dietas
Para o estudo, os pesquisadores internaram 20 adultos saudáveis sem diagnóstico de diabetes por quatro semanas seguidas no NIH Clinical Center. A média de idade dos participantes era de 29,9 anos e o índice de massa corporal (IMC) médio foi de 27,8 kg/m2.
Os participantes foram randomizados para consumir sem restrições alimentos à base de plantas, com baixo teor de gordura (10,3% de gordura, 75,2% de carboidratos) e com baixa densidade de energia (~ 1 kcal g-1) ou para consumir alimentos com baixo teor de carboidratos, proteína/gordura animal (75,8% de gordura, 10,0% de carboidratos) e alta densidade de energia (~ 2 kcal g-1) por duas semanas. Ao final deste período, os grupos foram cruzados e passaram a seguir a outra dieta por duas semanas.
As duas dietas continham cerca de 14% de proteína e foram pareadas pelo total de calorias, embora a dieta cetogênica tivesse duas vezes mais calorias por grama de alimento do que a dieta com baixo teor de gordura. Os participantes podiam comer o que e a quantidade que quisessem das refeições recebidas.
Um participante saiu do estudo devido a ocorrência de hipoglicemia na fase da dieta cetogênica. Para o desfecho primário, os pesquisadores compararam a ingestão média diária de energia sem restrições em cada período de duas semanas de dieta.
Eles observaram que a ingestão de energia da dieta com baixo teor de gordura foi menor em aproximadamente 550 a 700 kcal d-1, em comparação com a dieta cetogênica. No entanto, apesar das grandes diferenças na ingestão de calorias, os participantes não relataram diferenças na fome, prazer nas refeições ou saciedade entre as duas dietas.
Os participantes perderam peso com ambas (cerca de 1 kg a 2 kg em média), mas apenas a dieta com baixo teor de gordura levou a uma perda de gordura corporal significativa.
“Curiosamente, nossas descobertas sugerem benefícios para ambas as dietas, pelo menos em curto prazo”, disse o Dr. Kevin em um comunicado à imprensa.
“Embora a dieta vegetariana e com baixo teor de gordura ajude a reduzir a fome, a dieta cetogênica baseada no consumo de proteína/gordura animal resultou em níveis mais baixos e estáveis de insulina e glicose. Ainda não sabemos se essas diferenças seriam mantidas em longo prazo”, disse ele.
O Dr. Kevin ressaltou que o estudo não foi desenhado para fazer recomendações de dieta para perda ponderal, e os resultados poderiam ter sido diferentes se os participantes estivessem tentando ativamente perder peso.
Na verdade, eles nem sabiam do que se tratava o estudo; apenas dissemos que queríamos que seguissem as duas dietas para vermos o que aconteceria com o corpo deles quando comessem o quanto quisessem, disse ele ao Medscape.
“É difícil definir qual dieta pode ser melhor para um indivíduo. Eu acho que você pode interpretar este estudo como existindo pontos positivos e negativos para as duas dietas”, disse o Dr. Kevin.
“Tribos” das dietas
Comentando sobre o estudo para o Medscape, o Dr. Taylor Wallace, Ph.D., professor-adjunto do Departamento de Nutrição e Estudos Alimentares na George Mason University, nos EUA, disse que é importante ressaltar que “‘dieta com baixo teor de carboidratos’ ainda precisa ser definida, e existem muitas definições”.
“Nós realmente precisamos de uma definição padronizada do que constitui ‘baixo teor de carboidratos’, para que os estudos possam ser planejados e avaliados de uma forma consistente. É problemático, porque sem uma definição padrão, os pesquisadores da ‘tribo da dieta’ (cetogênica versus vegetariana) parecem sempre encontrar a resposta a seu favor”, disse o Dr. Taylor. “Este estudo parece usar menos de 20 gramas de carboidratos por dia, o que, na minha opinião, é um teor muito baixo de carboidratos.”
Talvez a ressalva mais importante, ele acrescentou, seja que no mundo real, “a maioria das pessoas não adere a dietas muito rigorosas – nem mesmo por duas semanas”.
O estudo foi financiado pelo NIDDK Intramural Research Program, com o apoio adicional do NIH de uma subvenção do National Institute of Nursing Research. Um autor recebeu remuneração por palestrar em conferências patrocinadas por fabricantes de produtos nutricionais, atua no conselho consultivo científico da Kerry Taste and Nutrition e faz parte de um consórcio acadêmico que recebeu financiamento para pesquisa da Abbott Nutrition, Nestec e Danone. O Dr. Kevin Hall e os outros autores informaram não ter conflitos de interesses relevantes. O Dr. Taylor Wallace é diretor e CEO do Think Healthy Group; editor do Journal of Dietary Supplements; e editor adjunto do Journal of the American College of Nutrition.
Nat Med. Publicado on-line em 21 de janeiro de 2021. Abstract
Medscape Notícias Médicas © 2021 WebMD, LLC
Citar este artigo: Dieta vegetariana ou cetogênica? – Medscape – 11 de fevereiro de 2021.
Deficiência de #vitamina D e Covid-19: o que há de novo?

Durante a pandemia, muito se questionou se haveria alguma relação entre a deficiência de vitamina D e Covid-19, principalmente em casos mais graves da infecção. A vitamina D é um hormônio esteroide responsável pela regulação dos níveis corporais de cálcio e sódio e da mineralização óssea. Também exerce importante papel no controle de diversas patologias, como doenças autoimunes e imunossupressão.
A vitamina D é sintetizada no organismo humano pela ingestão de alimentos ricos em proteínas animais e vegetais e pode ser classificada como colecalciferol (D3) e ergocalciferol (D2) respectivamente de acordo com sua origem alimentar. Além disso, é sintetizada pela exposição aos raios ultravioletas (UVB) que através da epiderme transforma um derivado do colesterol (7-DHC) em colecalciferol. No fígado, este é convertido em 25 hidroxi vitamina D que é transformada no seu produto final no rim. A maioria da população adquire vitamina D pela exposição solar, porém a população americana tem seu aporte mais relacionado à alimentação como leite, sucos e cereais fortificados com vitamina D. Populações que habitam altas altitudes e com maior concentração de melanina possuem deficiência natural da vitamina por estarem menos expostas à luz solar.
Uma análise com 4962 pacientes na National Health and Nutrition Examination Survey mostrou que quase 40% da amostra apresentava níveis de vitamina D abaixo de 20ng/ml e estavam mais relacionados à pessoas obesas e com diabetes tipo 1 e 2.
Deficiência de vitamina D e Covid-19
Atualmente alguns estudos vêm correlacionando a deficiência de vitamina D com um maior risco de complicações junto a infecção por Covid-19, principalmente em pacientes obesos, negros e com diabetes tipo 1 e 2. Porém, os estudos têm se mostrado confusos e esparsos como alguns exemplos citados abaixo:
- Um estudo realizado na França com 77 pacientes idosos e diagnosticados com Covid-19, evidenciou que aqueles que consumiam rotineiramente suplementação de vitamina D antes de serem infectados, apresentaram quadros mais leves em comparação com aqueles que tomaram vitamina D logo após o diagnóstico. E um estudo espanhol piloto randomizado com 76 pacientes hospitalizados com Covid-19, que receberam altas doses de vitamina D durante a internação, apresentaram menor incidência de internação em unidade fechada.
- Estudo em um hospital italiano mostrou uma ligação de baixos níveis de vitamina D com um risco aumentado de desenvolvimento de pneumonia por SARS CoV-2. Porém, idade, sexo e comorbidades apresentaram-se como maiores fatores de risco para mortalidade do que a deficiência de vitamina.
- Outro estudo realizado por pesquisadores do JAMA Network Open by University of Chicago observou uma grande conexão de deficiência de vitamina D com maiores chances de testagem positiva para Covid-19.
- Estudo realizado em Julho de 2020 na Indonésia, apesar de não ter sido revisto e publicado, tomou um grande vulto nas redes sociais de forma não oficial, também demonstrando uma forte ligação entre deficiência de vitamina D e maior risco de complicações por Covid-19. Porém, não foi possível analisar nenhum parâmetro desse estudo, o que o tornou desconsiderado.
- Em outubro de 2020, editores do PLoS One, também expressaram uma certa ligação entre níveis deficientes de vitamina D (abaixo de 30ng/ml) com o dobro de chances de óbito por Covid-19.
Todos os estudos demonstrados apresentaram muitas variáveis e bastante conflito de interesses, principalmente relacionados à indústria de suplementação, o que impede que haja uma conclusão assertiva que possa correlacionar efetivamente a deficiência de vitamina D com casos graves de Covid-19. Porém, níveis adequados de vitamina D são essenciais para a saúde óssea e a prevenção da deficiência vitamínica é sempre um objetivo clínico essencial.
Autor(a):
Gabriela Queiroz
Graduação em Medicina pela Universidade do Estado do Rio de Janeiro (UERJ) ⦁ Pós-Graduação em Anestesiologia pelo Ministério da Educação (MEC) ⦁ Pós-Graduação em Anestesiologia pelo Centro de Especialização e Treinamento da Sociedade Brasileira de Anestesiologia (CET/SBA) ⦁ Membro da Sociedade Brasileira de Anestesiologia (SBA) ⦁ Ênfase em cirurgias de trauma e emergência, obstetrícia, plástica estética reconstrutiva e reparadora e procedimentos endoscópicos ⦁ Experiência em trauma e cirurgias de emergência de grande porte, como ortopedia, vascular e neurocirurgia ⦁ Experiência em treinamento acadêmico e liderança de grupos em ambiente cirúrgico hospitalar ⦁ Orientadora acadêmica junto à classe de residentes em Anestesiologia ⦁ Orientadora e auxiliar em palestras regionais e internacionais na área de Anestesiologia.
Referências bibliográficas:
- Rubin R. Sorting Out Whether Vitamin D Deficiency Raises COVID-19 Risk.JAMA. 2021;325(4):329-330.
La exposición prenatal a #terapia antirretroviral puede contribuir a los cambios cardiacos en los bebés

NUEVA YORK, USA. Los bebés no infectados con virus de inmunodeficiencia humana expuestos a la terapia antirretroviral antes del nacimiento pueden tener cambios cardiovasculares “relevantes”, informan médicos en España.[1]
“Se observó deterioro cardiaco subclínico junto con presión arterial más alta y grosor de la íntima media carotídea más grueso en bebés expuestos al virus de inmunodeficiencia humana a los seis meses de edad. La mitad de ellos presentaba hipertensión”, informaron la Dra. Marta López Carbonell y sus colaboradores, de la Universidad de Barcelona, en Barcelona, España.
“Nuestros hallazgos respaldan posible aumento del riesgo cardiovascular en bebés no infectados por el virus de inmunodeficiencia humana que estuvieron expuestos en el útero a terapia antirretroviral”, escribieron en Clinical Infectious Diseases.
Los investigadores dieron seguimiento a 34 bebés no infectados expuestos al virus de inmunodeficiencia humana y a 53 bebés no expuestos hasta los 6 meses de edad. Los esquemas de terapia antirretroviral durante el embarazo incluyeron dos inhibidores nucleosídicos de la transcriptasa inversa (abacavir más lamivudina: 32%; emtricitabina más tenofovir: 41%, y zidovudina más lamivudina: 21%).
A la edad de 6 meses los lactantes no infectados expuestos al virus de inmunodeficiencia humana tenían paredes miocárdicas más gruesas (grosor medio de la pared septal: 5,02 frente a 3,98 mm; p < 0,001) y disfunción sistólica relativa con disminución del desplazamiento del anillo mitral (8,57 frente a 10,34 mm; p = 0,002 ) y disminución de tricúspide S’ (9,71 frente a 11,54 cm/s; p = 0,003) junto con disfunción diastólica relativa indicada por un tiempo prolongado de relajación isovolumétrica izquierda (58,57 frente a 47,94 ms; p < 0,001).
La evaluación vascular reveló presiones arteriales sistólicas (102 frente a 80 mm Hg; p < 0,001) y diastólicas (64 frente a 55 mm Hg; p = 0,045) significativamente más altas. La mitad de los niños expuestos al virus de inmunodeficiencia humana cumplió los criterios de hipertensión, en comparación con 3,77% del grupo no expuesto (p < 0,001).
Los niños expuestos al virus de inmunodeficiencia humana también tenían grosor de la íntima media carotídea medio más grueso (0,62 frente a 0,51 µm; p = 0,015).
“Estos cambios podrían permitirnos distinguirlos como población en riesgo incluso desde la vida fetal, estableciendo seguimiento estricto y más prolongado”, indicaron los autores.https://8726cd39b10dfee9ce799a8c59060cc7.safeframe.googlesyndication.com/safeframe/1-0-37/html/container.html
“Curiosamente, nuestro estudio muestra por primera vez que la hipertensión infantil podría predecirse a partir de la vida fetal mediante la evaluación de los esquemas maternos de terapia antirretroviral que contienen zidovudina o midiendo el espesor de la pared del tabique fetal mediante ecocardiografía fetal”, añadieron.
Los investigadores agregaron que sus hallazgos también “respaldan la tendencia actual de considerar los esquemas de terapia antirretroviral que contienen zidovudina durante el embarazo como alternativa, debido a la dosificación compleja y la asociación con tasas más altas de efectos adversos leves a moderados”.
Por lo tanto, “evitar los esquemas de terapia antirretroviral que contienen zidovudina durante el embarazo, promover hábitos de estilo de vida saludable y evitar otros riesgos cardiovasculares desde la niñez, podrían potencialmente prevenir eventos cardiovasculares más adelante en la vida”.
El estudio no tuvo financiamiento comercial. Los autores han declarado no tener ningún conflicto de interés económico pertinente. La Dra. López no respondió a una solicitud de comentarios al cierre de la edición.
Traducido y adaptado por el equipo de Medscape en español.
Reuters Health Information © 2021
Citar este artículo: La exposición prenatal a terapia antirretroviral puede contribuir a los cambios cardiacos en los bebés – Medscape – 9 de feb de 2021.
Una base de datos recoge más de 600 #productos sin gluten donde se valora su composición nutricional
Según la universidad, se trata del mayor análisis comparativo respecto a su composición, tanto en ingredientes como aspecto nutricional.

El grupo de investigación Alimentación y nutrición en la promoción de la salud de la Universidad CEU San Pablo, de Madrid, ha elaborado una base de datos con 629 alimentos sin gluten, de base cereal, disponibles en el mercado español, que, hasta la fecha, “representa el mayor análisis comparativo respecto a su composición, tanto en ingredientes como nutricional, con datos tomados a partir del etiquetado”, dice la propia universidad.
Elena Alonso Aperte, vicedecana de la Facultad de Farmacia del CEU y directora de este grupo, explica a CF que desde hace tiempo tienen abierta una línea de investigación centrada en “la valoración del estado nutricional de distintos colectivos de población y en el análisis de valor nutricional de los productos”. Uno de esos colectivos es el de los celiacos, que según Alonso Aperte, “tiene ciertas dificultades a la hora de seguir una dieta sin gluten”, que es el único tratamiento que tiene esta enfermedad. Así, recuerda que ya hicieron un trabajo en niños, de los cuales valoraron su dieta, estado nutricional, determinaciones bioquímicas y grasa corporal. “Entonces vimos que no hay datos de la composición de los productos sin gluten y decidimos investigar e hicimos esta base de datos con más de 600 productos sin gluten de base cereal de los que investigamos su composición. Y su composición no es fácil de conocer”.
Según la experta, esta base de datos puede servir de base para la realización de otros estudios sobre celiaquía.

La mayoría de los 629 productos alimenticios sin gluten considerados en el trabajo, realizado durante el año 2019, se incluyeron en las categorías de galletas, tartas y dulces (36,4%), seguido de pan y productos similares (24,2%) y pasta y productos similares (14%).
En su análisis, los investigadores vieron que existe una notable falta de datos en estos alimentos acerca de su composición nutricional, principalmente de su contenido en micronutrientes, como vitaminas y minerales. Hecho que justifica la necesidad de proporcionar nuevos datos en este sentido, para completar etiquetas, tablas o bases de datos de composición de alimentos.
A juicio de Alonso Aperte, esta carencia de información “no es algo intencionado de la industria alimentaria”. La razón es de otra índole. Y es que en las bases de datos sobre composición de los alimentos elaboradas por científicos (de donde se puede extraer esta información y entre las que destacan la de Moreiras y colaboradores y la base de datos on line Bedca) no se suelen incluir los alimentos especiales, como los que no tienen gluten. “Hacer estas bases de datos es algo es muy complejo”, reconoce la vicedecana de Farmacia.
Por otra parte, recuerda que la segunda fuente de información sobre composición nutricional de los productos, es el etiquetado. La ley dice que la industria solo está obligada a declarar en el etiquetado aquellos nutrientes “más importantes y con más incidencia en la salud, como la energía, las grasas, las grasas saturadas, las proteínas, los hidratos de carbonos, azúcares y sal“. “Estos los declaran, pero el resto de componentes son voluntarios”, afirma; “además, tampoco se puede declarar todo porque sería un etiquetado muy complicado”.
Suplementación o reforzar alimentos
Para Alonso Aperte, “es prioritario proporcionar estos datos a fin de poder llevar a cabo una correcta evaluación nutricional de la población celiaca en España”. Y es que, aunque aclara que la dieta de este colectivo sigue los mismos patrones que la de colectivos sanos, los celiacos presentan mayor riesgo de deficiencia en vitamina D, calcio, magnesio, hierro y ácido fólico, de ahí la importancia de conocer qué cantidades de estos micronutrientes llevan los productos sin gluten que consumen.
La solución a estas carencias pasaría a su juicio por dos medidas: la toma de suplementos alimenticios, “siempre que haya un diagnóstico previo de esas carencias para recomendarlos y pautar la suplementación adecuada” y mejorar la composición de los alimentos, “de tal manera que se pueda mejorar la ingesta de la vitamina o mineral de interés sin necesidad de un diagnóstico previo y llegando a todos los celiacos”. “Por ejemplo -continúa- añadir ácido fólico a los alimentos es una práctica muy común en algunos países, como en Estados Unidos, donde desde 1998 es obligatorio añadir ácido fólico a los productos de base cereal, con el fin de garantizar la ingesta de ácido fólico en las embarazadas para prevenir las malformaciones congénitas del tipo defectos del tubo neural. Es una fortificación que está muy ensayada y muy segura. Por tanto, si al alimento añades una cantidad que tiene que estimarse sobre la evidencia científica de esa vitamina, que normalmente no es tanto como la suplementación, sí consigues llegar a todos los celiacos a través de los alimentos y prevenir las situaciones de riesgo”.
Calidad del perfil nutricional, a debate
Otro debate abierto es sobre la calidad del perfil nutricional de los productos sin gluten. Sobre esta cuestión el trabajo del CEU arroja que gran parte de estos alimentos mostraron contenidos muy altos de energía (33,5%), grasas (28,5%), ácidos grasos saturados (30,0%), azúcares (21,6%) y sal (28,3%). Los productos estudiados se componían principalmente de harina de arroz y/o harina de maíz, y el 90% de ellos incluían almidón de arroz añadido.
La grasa añadida más común fue el aceite de girasol (presente en un tercio de los productos), seguido de lagrasa de palma, el aceite de oliva y el cacao. Solo el 24,5% de los productos tenía la declaración nutricional sin azúcar añadido. El 56% por ciento de los productos sin gluten empleaban la sacarosa en su formulación.
No obstante, el análisis de macronutrientes reveló que el 25,4% de los productos podrían etiquetarse como fuente de fibra. Además, en comparación con el estudio realizado en 2016, empieza a apreciarse una tímida reformulación en la composición de grasas y la reducción de sal, pero un menor uso de harinas alternativas y pseudocereales, que presentan un mejor perfil nutricional.
Más oferta y variedad
Para la experta, en España la industria alimentaria ha mejorado su oferta de productos sin gluten aptos para celiacos, mejorando aspectos como la variedad y la accesibilidad, dos viejas reivindicaciones del colectivo celiaco. Según sus datos, en 2017 España fue líder en aumentar su producción de alimentos sin gluten (18,8%), en comparación con Europa occidental (13,6%) y el resto del mundo (15,4%), siendo el tercer productor mundial de este tipo de productos, después de Estados Unidos y Brasil.
Las razones de este crecimiento no se deben solo al consumo de los productos sin gluten por parte de los celiacos, condición que alcanza al 1% de la población general del mundo occidental, también es impulsado por cambios en las actitudes del consumidor hacia la salud, como la tendencia a consumir alimentos libres de.
Para la experta, hay que tener cuidado con estas modas de consumir alimentos sin gluten cuando no hay un diagnóstico de celiaquía. Y es que eso puede tener un impacto negativo en la salud y en el bolsillo del consumidor. Así, Alonso Aperte asegura que “hacer una dieta sin gluten es complicado porque hay que eliminar productos y es difícil equilibrar la dieta. Por otra parte, puede enmascarar y retrasar el diagnóstico de una celiaquía; es decir si hay una alteración intestinal y se somete a una dieta sin gluten sin que haya una recomendación médica no se va a poder diagnosticar la enfermedad celiaca. Lo mismo ocurre con los que toman productos sin latosa”. Respecto al impacto económico, recuerda que los productos sin gluten son algo más caros que sus análogos con gluten, algo que también ha criticado el colectivo de celiacos.
Otros autores
Natalia Úbeda, codirectora del proyecto, y las profesoras Violeta Fajardo, Purificación González, Lourdes Samaniego y María Achón, junto con María Martínez completan la autoría de este trabajo
- ← Anterior
- 1
- 2
- 3
- …
- 860
- Próximo →